In situ solvent and counteranion-induced synthesis, structural characterization and photoluminescence properties of Pb-based MOFs†
Abstract
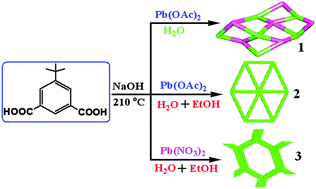
Based on an attempt to investigate the influence of solvents and counteranions on the structures and photoluminescence properties of Pb-based metal–organic frameworks, the hydrothermal reactions of the same amounts of Pb(NO3)2 and Pb(OAc)2 with the ligand 5-tert-butylisophthalic acid (H2tip) in the presence of water and a water–ethanol mixture have yielded three compounds [Pb(H2O)(tip)]n (1), [Pb3(μ4-O)(tip)2]n (2) and [Pb4(μ4-O)(tip)3]n (3) under similar reaction conditions (tip = 5-tert-butylisophthalate anion). These compounds represented the first examples of Pb(II) metal–organic frameworks with H2tip. All of them have been characterized by means of FT-IR spectra, elemental analysis, single-crystal X-ray diffraction, powder X-ray diffraction, thermogravimetric analysis, and photoluminescence spectra. The reaction processes revealed that 1 and 2 had selectivity for specific solvents. And the selectivity for specific counteranions was also found in 2 and 3. Single-crystal X-ray diffraction showed that 1 and 3 crystallized in the orthorhombic crystal system with space groups of Iba2 and Pna21, respectively, while 2 crystallized in the monoclinic P21/c space group. The structure of 1 featured a 2D bilayer structure containing a uninodal sql-type topological motif with a Schlafli symbol of (44). The 2D layer framework of 2 was constructed from a unique 8-connected hexanuclear cluster secondary building unit Pb6O2(COO)8, resulting in a uninodal hxl-type topological motif with a Schlafli symbol of (36·46·53). The structure of 3 could be described as a 3D microporous framework with a 6-connected tetranuclear cluster Pb4O(COO)6. Topological analysis revealed that 3 represented a uninodal dia-type topological motif with a Schlafli symbol of (66). All of the solid state compounds exhibited the photoluminescence properties at room temperature. Furthermore, taking the emissions of the free ligand into consideration, the emissions 1 and 2 could be assigned to metal-centered s → p transition transitions, while the emissions of 3 were due to ligand-to-metal charge transfers between the delocalized p bonds of carboxylate groups and p orbitals of Pb(II) centers. Especially, the in situ solvent and counteranion-induced synthesis strategy reported here could afford us new opportunities in the rapid design of new materials with interesting structures and properties.

 Please wait while we load your content...
Please wait while we load your content...