Tripodal tris(hydroxypyridinone) ligands for immunoconjugate PET imaging with 89Zr4+: comparison with desferrioxamine-B†
Abstract
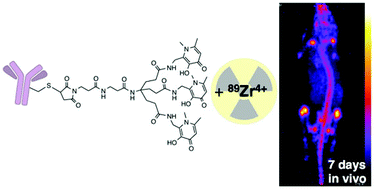
Due to its long half-life (78 h) and decay properties (77% electron capture, 23% β+, Emax = 897 keV, Eav = 397 keV, Eγ = 909 keV, Iγ = 100%) 89Zr is an appealing radionuclide for immunoPET imaging with whole IgG antibodies. Derivatives of the siderophore desferrioxamine-B (H3DFO) are the most widely used bifunctional chelators for coordination of 89Zr4+ because the radiolabeling of the resulting immunoconjugates is rapid under mild conditions. 89Zr-DFO complexes are reportedly stable in vitro but there is evidence that 89Zr4+ is released in vivo, and subsequently taken up by the skeleton. We have evaluated a novel tripodal tris(hydroxypyridinone) chelator, H3CP256 and its bifunctional maleimide derivative, H3YM103, for coordination of Zr4+ and compared the NMR spectra, and the 89Zr4+ radiolabeling, antibody conjugation, serum stability and in vivo distribution of radiolabelled immunoconjugates with those of H3DFO and its analogues. H3CP256 coordinates 89Zr4+ at carrier-free concentrations forming [89Zr(CP256)]+. Both H3DFO and H3CP256 were efficiently radiolabelled using [89Zr(C2O4)4]4− at ambient temperature in quantitative yield at pH 6–7 at millimolar concentrations of chelator. Competition experiments demonstrate that 89Zr4+ dissociates from [89Zr(DFO)]+ in the presence of one equivalent of H3CP256 (relative to H3DFO) at pH 6–7, resulting largely in [89Zr(CP256)]+. To assess the stability of H3DFO and H3YM103 immunoconjugates radiolabelled with 89Zr, maleimide derivatives of the chelators were conjugated to the monoclonal antibody trastuzumab via reduced cysteine side chains. Both immunoconjugates were labelled with 89Zr4+ in >98% yield at high specific activities and the labeled immunoconjugates were stable in serum with respect to dissociation of the radiometal. In vivo studies in mice indicate that 89Zr4+ dissociates from YM103-trastuzumab with significant amounts of activity becoming associated with bones and joints (25.88 ± 0.58% ID g−1 7 days post-injection). In contrast, <8% ID g−1 of 89Zr activity becomes associated with bone in animals administered 89Zr-DFO-trastuzumab over the course of 7 days. The tris(hydroxypyridinone) chelator, H3CP256, coordinates 89Zr4+ rapidly under mild conditions, but the 89Zr-labelled immunoconjugate, 89Zr-YM103-trastuzumab was observed to release appreciable amounts of 89Zr4+in vivo, demonstrating inferior stability when compared with 89Zr-DFO-trastuzumab. The significantly lower in vivo stability is likely to be a result of lower kinetic stability of the Zr4+ tris(hydroxypyridinone complex) relative to that of DFO and its derivatives.
- This article is part of the themed collection: Dalton Discussion 15: Metal ions in medical imaging

 Please wait while we load your content...
Please wait while we load your content...