Synthesis and reactivity of coordinatively unsaturated halocarbonyl molybdenum PNP pincer complexes†
Abstract
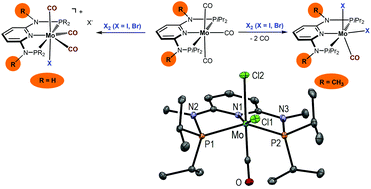
In the present study a series of six-coordinate neutral 16e halocarbonyl Mo(II) complexes of the type [Mo(PNPMe-iPr)(CO)X2] (X = I, Br, Cl), featuring the PNP pincer ligand N,N′-bis(diisopropylphosphino)-N,N′-dimethyl-2,6-diaminopyridine (PNPMe-iPr), were prepared and fully characterized. The synthesis of these complexes was accomplished by different methodologies depending on the halide ligands. For X = I and Br, [Mo(PNPMe-iPr)(CO)I2] and [Mo(PNPMe-iPr)(CO)Br2] were obtained by reacting [Mo(PNPMe-iPr)(CO)3] with stoichiometric amounts of I2 and Br2, respectively. In the case of X = Cl, [Mo(PNPMe-iPr)(CO)Cl2] was afforded by the reaction of [Mo(CO)4(μ-Cl)Cl]2 with 1 equiv. of PNPMe-iPr. The equivalent procedure also worked for X = Br. The modification of the 2,6-diaminopyridine scaffold by introducing NMe instead of NH spacers between the aromatic pyridine ring and the phosphine moieties changed the steric properties of the PNP-iPr ligand significantly. While in the present case exclusively neutral six-coordinate complexes of the type [Mo(PNPMe-iPr)(CO)X2] were obtained, with the parent PNP-iPr ligand, i.e. featuring NH spacers, cationic seven-coordinate complexes of the type [Mo(PNP-iPr)(CO)3X]X were afforded. Upon treatment of [Mo(PNPMe-iPr)(CO)X2] (X = Br, Cl) with Ag+ in CH3CN, the cationic complexes [Mo(PNPMe-iPr)(CO)(CH3CN)X]+ were formed. Halide abstraction from [Mo(PNPMe-iPr)(CO)Cl2] in THF–CH2Cl2 afforded [Mo(PNPMe-iPr)(CO)(THF)Cl]+. In keeping with the facile synthesis of monocationic complexes preliminary ESI-MS and DFT/B3LYP studies revealed that one halide ligand in complexes [Mo(PNPMe-iPr)(CO)X2] is labile forming cationic fragments [Mo(PNPMe-iPr)(CO)X]+ which react with molecular oxygen in parallel pathways to yield mono and dioxo Mo(IV) and Mo(VI) species. Structures of representative complexes were determined by X-ray single crystal analyses.

 Please wait while we load your content...
Please wait while we load your content...