EXAFS simulation refinement based on broken-symmetry DFT geometries for the Mn(iv)–Fe(iii) center of class I RNR from Chlamydia trachomatis
Abstract
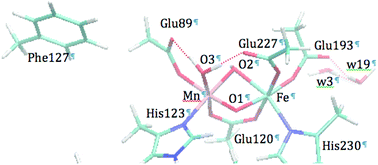
Ribonucleotide reductases (RNRs) catalyze the reduction of ribonucleotides into deoxyribonucleotides necessary for DNA biosynthesis. Unlike the conventional class Ia RNRs which use a diiron cofactor in their subunit R2, the active site of the RNR-R2 from Chlamydia trachomatis (Ct) contains a Mn/Fe cofactor. The detailed structure of the Mn/Fe core has yet to be established. In this paper we evaluate six different structural models of the Ct RNR active site in the Mn(IV)/Fe(III) state by using Mössbauer parameter calculations and simulations of Mn/Fe extended X-ray absorption fine structure (EXAFS) spectroscopy, and we identify a structure similar to a previously proposed DFT-optimized model that shows quantitative agreement with both EXAFS and Mössbauer spectroscopic data.

 Please wait while we load your content...
Please wait while we load your content...