Critical adsorption of polyelectrolytes onto charged Janus nanospheres
Abstract
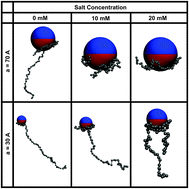
Based on extensive Monte Carlo simulations and analytical considerations we study the electrostatically driven adsorption of flexible polyelectrolyte chains onto charged Janus nanospheres. These net-neutral colloids are composed of two equally but oppositely charged hemispheres. The critical binding conditions for polyelectrolyte chains are analysed as function of the radius of the Janus particle and its surface charge density, as well as the salt concentration in the ambient solution. Specifically for the adsorption of finite-length polyelectrolyte chains onto Janus nanoparticles, we demonstrate that the critical adsorption conditions drastically differ when the size of the Janus particle or the screening length of the electrolyte are varied. We compare the scaling laws obtained for the adsorption–desorption threshold to the known results for uniformly charged spherical particles, observing significant disparities. We also contrast the changes to the polyelectrolyte chain conformations close to the surface of the Janus nanoparticles as compared to those for simple spherical particles. Finally, we discuss experimentally relevant physico-chemical systems for which our simulations results may become important. In particular, we observe similar trends with polyelectrolyte complexation with oppositely but heterogeneously charged proteins.

 Please wait while we load your content...
Please wait while we load your content...