Oxygen vacancy formation and the ion migration mechanism in layered perovskite (Sr,La)3Fe2O7−δ†
Abstract
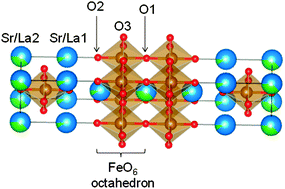
Metal oxides are widely used in devices such as sensors, fuel cells, and oxygen permeation membranes. Understanding the oxide ion migration mechanism would provide fundamental insights into the relationships between the structure and properties such as ionic conductivity. The Ruddlesden–Popper perovskite (Sr,La)n+1(Fe,Co)nO3n+1 (n = 2) has characteristic oxygen permeation and ion conduction properties, resulting from the layered perovskite structure. To elucidate the ion migration mechanism in Sr2.46La0.54Fe2O7−δ (SLF) we used a combination of experimental techniques [X-ray powder diffraction (XRPD) and enthalpy investigations of the oxygen vacancy formation reaction] and computational techniques [the bond valence sum (BVS) approach and ab initio density functional theory (DFT)]. The structural analyses of SLF by XRPD and DFT agreed well. They showed that the oxygen vacancies in SLF are located at the O1 oxygen site, which is on the vertex shared by two FeO6 octahedra in the perovskite layer. Enthalpy of the oxygen vacancy formation changed at 830 °C. This is similar to the ionic conduction behavior reported for Sr3Fe2O7. The XRPD study indicates that the host structural framework did not change with temperature, while the oxygen/vacancy arrangement in SLF did change at 830 °C. The BVS and DFT studies suggested a change in the ion migration pathway, in which the ion migration through O1 sites becomes more important at temperatures higher than 830 °C.

 Please wait while we load your content...
Please wait while we load your content...