Insight into band positions and inter-particle electron transfer dynamics between CdS nanoclusters and spatially isolated TiO2 dispersed in cubic MCM-48 mesoporous materials: a highly efficient system for photocatalytic hydrogen evolution under visible light illumination†
Abstract
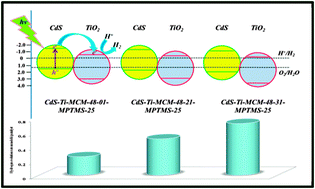
CdS incorporated Si-MCM-48 and Ti-MCM-48 cubic phased mesoporous photocatalysts were prepared by a two-step modification synthetic approach under relatively mild conditions. A highly efficient (24.8%, apparent quantum yield (AQY)) photocatalyst for visible light (λ > 400 nm) enabled solar hydrogen evolution can be realized by assembling CdS with Ti-MCM-48 cubic mesoporous materials in the absence of a noble metal co-catalyst. The photocatalytic mechanism was thoroughly investigated and demonstrated by conducting a wealth of characterization techniques such as powder X-ray diffraction (XRD), nitrogen adsorption isotherm, transmission electron microscopy (TEM), UV-visible diffuse reflectance spectroscopy (DRS), X-ray photoelectron spectroscopy (XPS), ultraviolet photoelectron spectroscopy (UVPS), atomic absorption spectroscopy (AAS), photoluminescence (PL) spectroscopy, time-resolved fluorescence emission decay, and electron paramagnetic resonance (EPR) spectroscopy studies. This work is the first to unambiguously identify the band positions of both CdS and TiO2 encapsulated in porous materials. The photocatalytic activity of the CdS incorporated Ti-MCM-48 mesoporous photocatalysts was found to be dependent on the content of both CdS and TiO2. A correlation between the electron injection efficiency and the photocatalytic activity was established as well in the CdS incorporated Ti-MCM-48 mesoporous photocatalysts.

 Please wait while we load your content...
Please wait while we load your content...