Crystal-to-crystal transformation in solid-state electrochemical doping of Cl− ions to the nanoporous neutral radical lithium phthalocyanine: revelation of electron–electron correlations in the 1D half-filled system†
Abstract
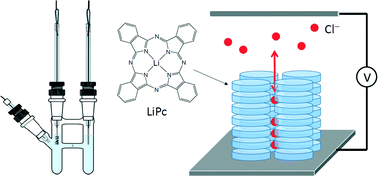
We performed electrochemical doping of Cl− ions to the x-form single crystals of a neutral π radical LiPc, in which the structure consists of 1D stacking chains of LiPc and nanochannels parallel to them. It was found that the LiPc crystals on the working electrode were electrochemically oxidized in an acetonitrile solution of tetrabutylammonium chloride (TBA·Cl), exhibiting a crystal-to-crystal transformation to LiPc·Clx (0 < x < 0.5), in which the x value was controlled by the electrochemical potential. The crystal structures of the LiPc·Clx series were successfully solved and refined by X-ray single-crystal analysis, and the obtained structures clearly indicated the presence of Cl− ions in the 1D channels and a systematic structural change in the π stacking chain of LiPc. As the value of x increased, the interplanar distance between the neighboring LiPc molecules decreased, which was associated with a significant enhancement of conductivity. These oxidative-doping effects indicate that the structural and electrical properties of the 1D half-filled electron system of LiPc are governed by electron–electron correlations between the unpaired electrons.

 Please wait while we load your content...
Please wait while we load your content...