Controlling the kinetics of the non-covalent functionalization of carbon nanotubes using sub-cmc dilutions in a co-surfactant environment†
Abstract
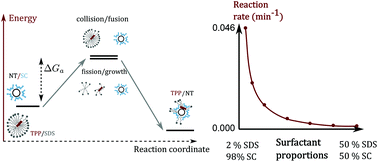
We investigate the origin of the slow kinetics of functionalization processes in micellar environments. We show that the ionic nature of the surfactants used to solubilize small molecules and nano-objects plays a central role in the slowness of the kinetics. In order to solve this issue, we have developed an innovative method that we apply to the hybrid compound porphyrin molecule/carbon nanotube. We use two ionic surfactants to solubilize the molecules and the nanotubes respectively. Passing the molecule suspension below the cmc allows circumventing the stability of the ionic surfactant while keeping the benefit of working with highly concentrated solutions. This method allows fine control of the functionalization reaction and tuning of the kinetics characteristic time over more than two orders of magnitude.


 Please wait while we load your content...
Please wait while we load your content...