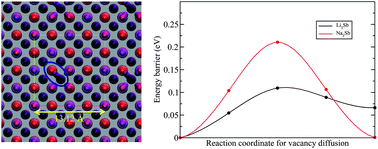
A detailed comparative study between the electrochemical lithiation and sodiation of pure antimony (Sb), relating changes in structural, thermodynamic, kinetic and electrochemical properties has been carried out. For this purpose, a wide range of measurements using electrochemical (galvanostatic cycling, GITT, PITT), X-ray diffraction (XRD) and X-ray photoelectron spectroscopy (XPS) methods as well as density functional theory (DFT) based investigations have been undertaken. Assessment of the thermodynamics reveals that the reaction proceeds identically during the first and second cycles for Li whereas it differs between the first and subsequent cycles for Na as the reaction with Na proceeds through a different pathway associated with the formation of amorphous NaxSb phases. For the first time we rationalize the amorphization of NaxSb phases by the long ranged strain propagation due to Na-vacancy compared to Li–Sb. At full discharge, our XRD results show for the first time that a minor fraction of hexagonal Li3Sb forms concomitantly with cubic Li3Sb. The XRD results confirm that Sb crystallizes into hexagonal Na3Sb at full sodiation. The kinetics of the reaction is assessed by rate performance tests which highlight that both Li and Na can diffuse rapidly throughout micron thick films at room temperature. However, it is found that the (de)insertion of Li provides lower overpotentials and larger storage capacities compared to Na. The difference in rate performance is complemented by diffusion coefficient determinations near the 0 V region where both materials are crystallized into M3Sb (M = Li, Na). Interestingly, calculations show that the energy barrier for near-neighbor vacancy migration, predominant in these close-packed phases, is about twice for Na than for Li. Our analysis tries to relate the lower intrinsic diffusivity of Na compared to Li with the long-range strain propagation induced by the former, thereby leading to an intrinsic origin of differences in rates, mechanical properties and amorphization. Finally, the surface chemistry of Sb electrodes cycled in NaClO4 dissolved in pure PC with(out) the addition of 5 wt% EC or FEC shows presence of ethers and NaF for the EC- and FEC-based electrolytes, respectively, and SEI films rich in Na-based carbonates.

 Please wait while we load your content...
Please wait while we load your content...