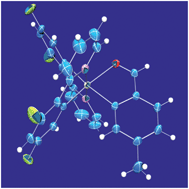
Cyclometalated iridium(III) complexes have been prepared in high yields from base-assisted transmetalation reactions of cis-bis(aquo)iridium(III) complexes with boronated aromatic proligands. Reactions proceed at room temperature. Potassium hydroxide and potassium phosphate are effective supporting bases. Kinetic, meridional isomers are isolated because of the mildness of the new technique. Syntheses are faster with KOH, but the gentler base K3PO4 broadens the reaction's scope. Complexes of chelated ketone, aldehyde, and alcohol complexes are reported that bind iridium through formally neutral oxygen and formally anionic carbon. The new complexes luminesce with microsecond-scale lifetimes at 77 K and nanosecond-scale lifetimes at room temperature; emission quenches in air. Two complexes, an aldehyde and its reduced (alcohol) derivative, are crystallographically characterized. Their bonding is examined with density-functional theory calculations. Time-dependent computations suggest that the Franck–Condon triplet states of these complexes have mixed orbital parentage, arising from one-particle transitions that mingle through configuration interaction.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...