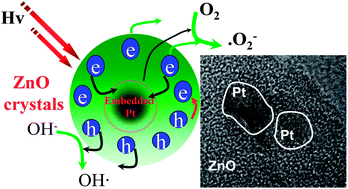
Noble metal/semiconductor nanocomposites play an important role in high efficient photocatalysis. Herein, we demonstrate a facile strategy for fabrication of hollow Pt-ZnO nanocomposite microspheres with hierarchical structure under mild solvothermal conditions using Zn (CH3COO)2·2H2O and HPtCl4 as the precursors, and polyethylene glycol-6000 (PEG-6000) and ethylene glycol as the reducing agent and solvent, respectively. The as-synthesized ZnO and Pt-ZnO composite nanocrystals were well characterized by powder X-ray diffraction (XRD), nitrogen-physical adsorption, scanning electron microscopy (SEM), energy dispersive X-ray (EDX), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), UV-vis diffuse reflectance spectra (DRS), and photoluminescence (PL) emission spectroscopy. It was found that Pt content greatly influences the morphology of Pt-ZnO composite nanocrystals. Suitable concentration of HPtCl4 in the reaction solution system can produce well hierarchically hollow Pt-ZnO nanocomposite microspheres, which are composed of an assembly of fine Pt-ZnO nanocrystals. Photocatalytic tests of the Pt-ZnO microspheres for the degradation of the dye acid orange II revealed extremely high photocatalytic activity and stability compared with those of pure ZnO and corresponding Pt deposited ZnO. The remarkable photocatalytic performance of hollow Pt-ZnO microspheres mainly originated from their unique nanostructures and the low recombination rate of the e−/h+ pairs by the platinum nanoparticles embedded in ZnO nanocrystals.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...