Novel FK506 and FK520 analogues via mutasynthesis: mutasynthon scope and product characteristics†
Abstract
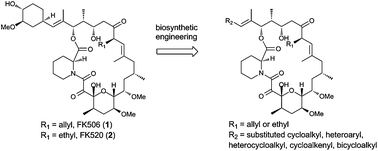
Novel FK506 and FK520 analogues were generated via biosynthetic engineering in order to generate analogue compounds with equal potency but improved pharmacological profiles compared to FK506. Strains suitable for mutasynthesis were produced by abolishing the activity of the rapK homolog fkbO, thereby disabling starter unit biosynthesis, and by replacing the
 Please wait while we load your content...
Please wait while we load your content...