Achieving high efficiency and cyclability in inexpensive soluble lead flow batteries†
Abstract
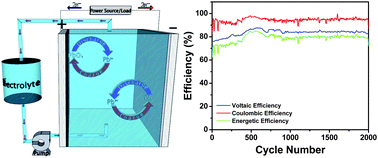
Here we report on the significant improvements made in the energy efficiency and cycle life of full-cell soluble lead flow batteries (SLFBs). We describe energy efficiency loss mechanisms, particularly in context to the deposition of PbO2 at the positive electrode. The morphology and crystal structure of deposits formed at the positive electrode, under galvanostatic and potentiostatic conditions, were characterized using both

 Please wait while we load your content...
Please wait while we load your content...