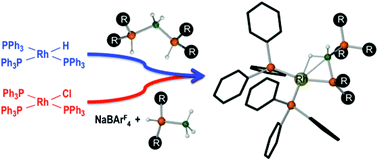
The synthesis of a range of bis(phosphine)boronium salts is reported [(R2HP)2BH2][X] (R = Ph, tBu, Cy) in which the counter anion is also varied (X− = Br−, [OTf]−, [BArF4]−, ArF = 3,5-(CF3)2C6H3). Characterization in the solid-state by X-ray diffraction suggests there are weak hydrogen bonds between the PH units of the boronium cation and the anion (X− = Br−, [OTf]−), while solution NMR spectroscopy also reveals hydrogen bonding occurs in the order [BArF4]− < [OTf]− < Br−. [(Ph2HP)2BH2][BArF4] reacts with RhH(PPh3)3, by elimination of H2, forming [Rh(κ1,η-PPh2BH2·PPh2H)(PPh3)2][BArF4] which shows a β-B-agostic interaction from the resulting base stabilised phosphino–borane ligand. Alternatively such ligands can be assembled directly on the metal centre by reaction of in situ generated {Rh(PPh3)3}+ and Ph2HP·BH3 to afford [Rh(κ1,η-PPh2BH2·PPh3)(PPh3)2][BArF4], which was characterised by X-ray crystallography. Addition of H3B·PPh2H to the well-defined 16-electron “T-shaped” complex [Rh(PiBu3)2(PPh3)][BArF4] (characterised by X-ray crystallography) formed of a mixture of base-stabilised phosphino borane ligated complexes [Rh(κ1,η-PR2BH2·PR3)(PR3)2][BArF4] (R = iBu or Ph). These last observations may lend clues to the formation of bis(phosphine)boronium salts in the catalytic dehydrocoupling reaction of phosphine boranes as mediated by Rh(I) compounds.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...