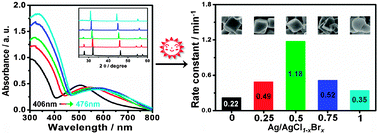
The solid solution (SS) method is an effective way to design impactful photocatalysts, owing to its merit of continuous bandgap-tuning. A calcination, usually breaking the morphology of a material, has to be used to synthesize such catalysts, although the morphology is a critical issue affecting its catalytic behavior. It thus is strongly desired to construct SS-based catalysts with a shaped morphology. Here, we report that AgCl1–xBrx SS-based photocatalysts, Ag–AgCl1–xBrx, with a shaped morphology, can be produced via an ion-exchange between nanostructured Ag–AgCl and KBr. It is found that when sphere-like Ag–AgCl is employed as a precursor, the Ag–AgCl1–xBrx, maintains its morphology when x is in the range of 0–1. The bandgap, and the catalytic activities of these Ag–AgCl1–xBrx for the degradation of methyl orange, display a monotonic narrowing and a continuous enhancement, respectively, with the increase of x. In contrast, when cube-like Ag–AgCl is used as a precursor, the Ag–AgCl1–xBrx preserves its morphological features when x ≤ 0.5, while a morphology distortion is observed when x ≥ 0.75. Fascinatingly, although the bandgap of thus-constructed Ag–AgCl1–xBrx also exhibits a monotonic narrowing with the increase of x, they (x ≠ 0, 1) display enhanced catalytic activity compared with the two terminal materials, Ag–AgCl and Ag–AgBr, wherein Ag–AgCl0.5Br0.5, with a cube-like morphology, shows the highest catalytic performance. The synergistic effect of morphology selection and bandgap narrowing plays an important role for these intriguing new findings. Our work provides a unique forum for an optimized selection of SS-based photocatalysts in terms of morphology selection and bandgap-tuning.

 Please wait while we load your content...
Please wait while we load your content...