Synthetic hydrogel platform for three-dimensional culture of embryonic stem cell-derived motor neurons†
Abstract
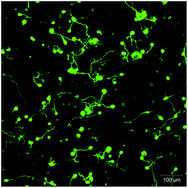
Culturing mammalian neurons in three-dimensional (3D) microenvironments that more closely recapitulate critical biochemical and biophysical aspects of the developing or adult central nervous system (CNS) milieu remains a significant challenge in neurobiological studies and in regenerative medicine. Here, we aimed to exploit recent advances in poly(ethylene glycol) (PEG) hydrogel chemistries to define a synthetic niche capable of supporting the culture and axonal outgrowth of both aggregated and dissociated mouse embryonic stem cell-derived motor neurons (ESMNs). Using
- This article is part of the themed collection: Biomaterials Science Lectureship Winners

 Please wait while we load your content...
Please wait while we load your content...