Iridium catalyzed enantioselective hydrogenation of α-alkoxy and β-alkoxy vinyl ethers†
Abstract
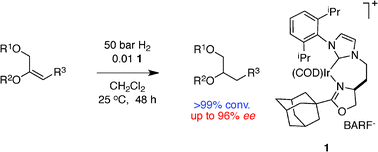
Iridium catalyzed, enantioselective hydrogenations of functionalized, acid-sensitive vinyl ethers are reported. These reactions are mediated by the chiral N-carbene–oxazoline complex 1 that, unlike similar N,P-ligated

 Please wait while we load your content...
Please wait while we load your content...