Porphyrin–DNA conjugates: porphyrin induced adenine–guanine homoduplex stabilization and interduplex assemblies†
Abstract
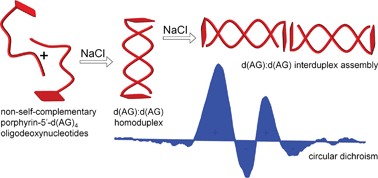
DNA has found widespread uses as a nanosized scaffold for assembly of patterned multichomophoric nanostructures. Herein we report the synthesis, self-assembly, stability, and spectroscopic studies of short alternating non-self-complementary DNA sequences 5′-(dGdA)4 and 5′-(dAdG)4 with non-charged tetraarylporphyrins covalently linked to the 5′ position of deoxyadenosine or deoxyguanosine via a phosphate or amide linker. The linker, the metal in the porphyrin coordination center, and the neighboring nucleobase have very distinct effects on the duplex formation of porphyrin–deoxyguanosine–deoxyadenosine oligodeoxynucleotides. At ionic strength between 5 mM and 40 mM, free base trispyridylphenylporphyrin appended to the 5′ termini of 5′-(dAdG)4 oligonucleotide via short non-polar amide linker served as a hydrophobic molecular cap inducing deoxyadenosine–deoxyguanosine antiparallel homoduplex. At ionic strength of ≥60 mM, the free base porphyrin functioned as a molecular ‘glue’ and induced the formation of porphyrin–DNA inter-homoduplex assemblies with characteristic tetrasignate CD Cotton effects in the porphyrin Soret band region. When the porphyrin cap was covalently attached to 5′ position of deoxyguanosine or deoxyadenosine via charged phosphate linker, no significant deoxyadenosine–deoxyguanosine hybridization was observed even at elevated ionic strengths.

 Please wait while we load your content...
Please wait while we load your content...