Surfactant free RGO/Pd nanocomposites as highly active heterogeneous catalysts for the hydrolytic dehydrogenation of ammonia borane for chemical hydrogen storage†
Abstract
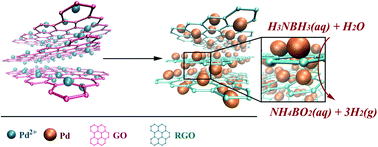
In this study, monodisperse palladium (Pd)
* Corresponding authors
a
Key Laboratory of Nonferrous Metal Chemistry and Resources Utilization of Gansu Province, State Key Laboratory of Applied Organic Chemistry and Colleague of Chemistry and Chemical Engineering, Lanzhou University, Lanzhou 730000, P. R. China
E-mail:
xipx@lzu.edu.cn, zengzhzh@lzu.edu.cn
Fax: +86 931 8912582
Tel: +86 931 8912589
b State Key Laboratory of Advanced Ceramic Fibers & Composites, College of Aerospace and Materials Engineering, National University of Defense Technology, Changsha, P. R. China
c Colleague of Chemistry and Chemical Engineering, Xuchang University, Xuchang 461000, P. R. China
d Univ Paris Diderot, Sorbonne Paris Cité, Unité de Biologie Fonctionnelle et Adaptative, CNRS EAC4413, Paris, France
In this study, monodisperse palladium (Pd)

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
P. Xi, F. Chen, G. Xie, C. Ma, H. Liu, C. Shao, J. Wang, Z. Xu, X. Xu and Z. Zeng, Nanoscale, 2012, 4, 5597 DOI: 10.1039/C2NR31010D
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
