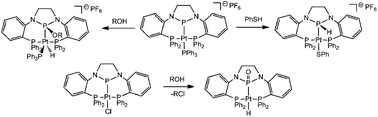
The reactivity of E–H bonds (E = S, O, Cl) with Pt(II) complexes ligated by an N-heterocyclic phosphido-containing diphosphine ligand have been investigated. Addition of PhSH to [(PPP)Pt(PPh3)][PF6] (1) results in clean formation of [(PP(H)P)Pt(SPh)][PF6] (3), in which the substrate has added across the Pt–PNHP bond. Similar reactivity occurs when 1 is treated with ROH (R = Ph, Me), but in this case the O–H bond adds across the Pt–P bond in the opposite direction producing [(PP(OR)P)Pt(H)(PPh3)][PF6] (R = Ph (4), Me (5)). HCl addition to 1 cleanly generates [(PP(H)P)PtCl][PF6] (6PF6PF6PF6). The neutral Pt–NHP complex (PPP)PtCl (2) exhibits similar reactivity; however, in the presence of the nucleophilic Cl− anion, the (PP(OR)P)Pt(H)Cl species presumably generated via addition of ROH (R = Me, Et) undergoes an Arbuzov-like dealkylation reaction to exclusively form the N-heterocylic phosphinito species (PP(O)P)Pt(H) (7).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...