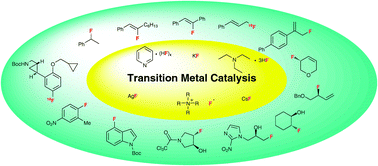
Transition metal catalyzed transformations using fluorinating reagents have been developed extensively for the preparation of synthetically valuable fluorinated targets. This is a topic of critical importance to facilitate laboratory and industrial chemical synthesis of fluorine containing pharmaceuticals and agrochemicals. Translation to 18F-radiochemistry is also emerging as a vibrant research field because functional imaging based on Positron Emission Tomography (PET) is increasingly used for both diagnosis and pharmaceutical development. This review summarizes how fluoride sources have been used for the catalytic nucleophilic fluorination of various substrates inclusive of aryl triflates, alkynes, allylic halides, allylic esters, allylic trichloroacetimidates, benzylic halides, tertiary alkyl halides and epoxides. Until recently, progress in this field of research has been slow in part because of the challenges associated with the dual reactivity profile of fluoride (nucleophile or base). Despite these difficulties, some remarkable breakthroughs have emerged. This includes the demonstration that Pd(0)/Pd(II)-catalyzed nucleophilic fluorination to access fluoroarenes from aryl triflates is feasible, and the first examples of Tsuji–Trost allylic alkylation with fluoride using either allyl chlorides or allyl precursors bearing O-leaving groups. More recently, allylic fluorides were also made accessible under iridium catalysis. Another reaction, which has been greatly improved based on careful mechanistic work, is the catalytic asymmetric hydrofluorination of meso epoxides. Notably, each individual transition metal catalyzed nucleophilic fluorination reported to date employs a different F-reagent, an observation indicating that this area of research will benefit from a larger pool of nucleophilic fluoride sources. In this context, a striking recent development is the successful design, synthesis and applications of a fluoride-derived electrophilic late stage fluorination reagent. This new class of reagents could greatly benefit preclinical and clinical PET imaging.

 Please wait while we load your content...
Please wait while we load your content...