Purinergic receptor
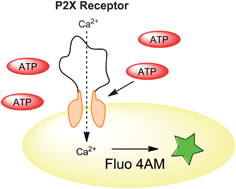
signaling events in platelets are a major determinant in platelet function. However, investigating the ATP-sensitive P2X1 platelet receptor is difficult due to its rapid desensitization in the washed platelet sample matrix. To minimize desensitization, most studies involving P2X1 activity in washed platelets require apyrase in the sample to reduce matrix ATP levels. Unfortunately, the apyrase will also rapidly degrade any ATP added exogenously during the studies. Here, we describe a method that employs the reported P2X1 inhibitor NF449 to sensitize washed platelets in the absence of any added apyrase. Sensitization is verified by spectrofluorometric determination of Ca2+ entry into the platelets after stimulation with concentrations of ATP ranging from 0.625 μM to 5 μM. Results suggest that sensitization of the P2X1 receptor by NF449 is not necessarily dependent upon the inhibitor concentration, but rather the ratio of the inhibitor to exogenously-added ATP concentrations. With a ratio of ATP agonist to NF440 concentration of ∼5 : 1, the resulting percent change in fluorescence due to Ca2+ entry into the platelet is 39.3 ± 0.8%; however, at a ratio of 1 : 8 ATP to NF449 the percent change is reduced to 13.1 ± 2.2%. The sensitizing effect is also investigated as a function of time. The results obtained verify that NF449 can behave as a concentration-dependent inhibitor and sensitizer of the plateletP2X1 receptor in washed platelet samples, depending on the ATP concentration in the matrix.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...