Supramolecular polymerization of oligopyrenotides – stereochemical control by single, natural nucleotides†‡
Abstract
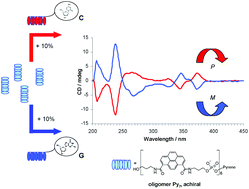
Amphiphilic heptapyrenotides (Py7) assemble into supramolecular
- This article is part of the themed collection: Nucleic acids: new life, new materials
 Please wait while we load your content...
Please wait while we load your content...