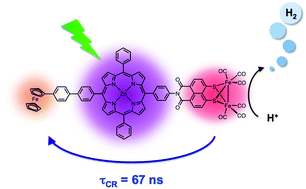
Two covalently linked linear electron donor–acceptor triads Fc-ZnTPP-[NMI-FeI-FeI-S2(CO)6] (1) and Fc-Ph-ZnTPP-[NMI-FeI-FeI-S2(CO)6] (2) consisting of a zinc meso-tetraphenylporphyrin (ZnTPP) chromophore, a naphthalene monoimide diiron hydrogenase active site model [NMI-FeI-FeI-S2(CO)6], and a ferrocene (Fc) secondary electron donor have been synthesized along with their corresponding dyad reference molecules ZnTPP-[NMI-FeI-FeI-S2(CO)6] (3), Fc-ZnTPP (4), and Fc-Ph-ZnTPP (5). Time-resolved transient absorption and emission studies in CH2Cl2 show that selective photoexcitation of ZnTPP in triads 1 and 2 results in two competing quenching pathways for 1*ZnTPP: electron transfer from 1*ZnTPP to [NMI-FeI-FeI-S2(CO)6] and energy transfer from 1*ZnTPP to low-lying Fc excited states. Our studies on reference dyads 4 and 5 show that the majority of 1*ZnTPP produced by the laser pulse decays rapidly by energy transfer to Fc in triad 1 (τ < 10 ps), while electron transfer to [NMI-FeI-FeI-S2(CO)6] dominates in triad 2, allowing the second rapid electron transfer step from Fc to ZnTPP+˙ to proceed. Quantum yields of the fully charge separated states Fc+-ZnTPP-[NMI-Fe0-FeI-S2(CO)6] and Fc+-Ph-ZnTPP-[NMI-Fe0-FeI-S2(CO)6] are 0.13 and 0.71, respectively. Charge recombination in Fc+-ZnTPP-[NMI-Fe0-FeI-S2(CO)6] occurs with τCR = 9 ± 1 ns and τCR = 67 ± 2 ns for Fc+-Ph-ZnTPP-[NMI-Fe0-FeI-S2(CO)6]. By incorporating a secondary electron donor, the lifetime of the reduced diironhydrogenase mimic was extended by a factor of >450. Studies of photochemical hydrogen evolution using 1 and 2 reveal that the hydrogen generation efficiency depends on the lifetime of the final charge separated state. The ability to execute a multi-electron proton-coupled electron transfer mechanism in a stepwise manner will allow us to investigate the structural and electronic requirements for each step aiding in overall system optimization. Thus, it is possible to use the same multi-step electron transfer strategy that has been employed to extend the lifetime of charge-separated states in photodriven donor–acceptor systems to extend the lifetime of the reduced states of metal complexes of potential use in catalytic proton reduction.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...