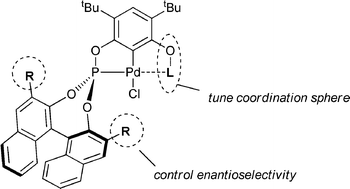
A range of chiral resorcinol bis(phosphite) and phosphite–phosphinite ligands were produced and their propensity to form palladiumPCP-pincer complexes examined. The ease of base-assisted C–H palladation of the ligands falls in the order bis(phosphinite) > phosphite–phosphinite > bis(phosphite). The catalytic activity of the complexes in the asymmetric allylation of benzaldehyde with allyl tributyltin was examined and it was found that, contrary to expectations, ligands with 3,3′-disubstituted BINOL residues show poorer activity and stereoselectivity than unsubstituted BINOL analogues. In addition the order of activity of the pincer complexes was established as bis(phosphite) > phosphite–phosphinite > bis(phosphinite). Crystal structures of representative examples of a 3,3′-disubstituted BINOL, mono- and bis(phosphite) ligands based on 2,4-di-tert-butyl resorcinol and Pd complexes of two of the chiral complexes are presented.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...