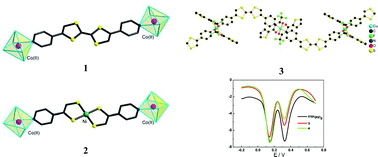
The bis(pyridyl)-substituted TTF derivative, 2,6(7)-bis(4-pyridyl)-1,4,5,8-tetrathiafulvalene (TTF(py)2), and an inorganic analogue, [Ni(4-pedt)2] (4-pedt = 1-(pyridin-4-yl)ethylene-1,2-dithiolate), were used as bridging ligands to construct two multinuclear complexes {CoII2(TpPh2)2(OAc)2[TTF(py)2]} (1, TpPh2 = hydridotri(3,5-diphenylpyrazol-1-yl)borate) and {CoII2(TpPh2)2(OAc)2[Ni(4-pedt)2]} (2), and two 1D zigzag chain complexes, {[MII(tta)2][TTF(py)2]}n (M = Cu for 3, and Mn for 4; tta = thenoyltrifluoroacetonate). X-Ray structural studies indicate that complexes 1 and 2 are very similar as a result of the isolobal analogy between TTF(py)2 and [Ni(4-pedt)2], whereas complexes 3 and 4 are isostructural. The absorption spectra, electrochemical and magnetic properties for these new complexes have been studied. The results show that the interactions between the paramagnetic ions are weak owing to the large separation of the bridging ligands of TTFs and the inorganic analogue.
 Please wait while we load your content...
Please wait while we load your content...