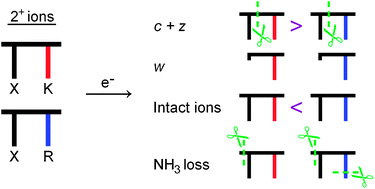
Electron capture by doubly charged peptide cations leads to neutral losses in addition to N–Cα bond cleavages that give c and z fragments. In this work we discuss the influence of amino acid sequence on hydrogen versus ammonia loss and the propensity for subsequent partial side-chain cleavage after ammonia loss to give w fragment ions. Experiments were done on two series of doubly protonated dipeptides, [XK+2H]2+ and [XR+2H]2+, where X is one of the twenty common amino acid residues, excluding aspartic acid (D), and K and R are lysine and arginine, respectively. While it was previously established that NH3 is lost exclusively from the N-terminal ammonium group and not from side-chain ammonium groups, we find here that ammonia can be lost from guanidinium radicals as well. The ratio between H loss and NH3 loss reveals some information on internal ionic hydrogen bonds and peptide conformation since proton sharing between the N-terminal ammonium group and a basic side chain decreases the probability for NH3 loss due to a lower recombination energy and as a result reduced capture probability. The abundance of w ions was found to correlate with the reaction energy for their formation; highest yield was found for CK and lowest for AK and HK. The survival rate of charge-reduced species was higher for XR than for XK, which is likely linked to the formation of long-lived Cα radicals in the latter case. The probability for N–Cα bond cleavage is smaller on average for XR than for XK which indicates that hydrogen transfer from the ε-ammonium radical to the amide group triggers some of the cleavages, or is a result of the different distances between the amide group and the charges in XR and XK. Finally, our data support the previous concept that charge partitioning between c and z fragments can be explained by competition between the two fragments for the proton.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...