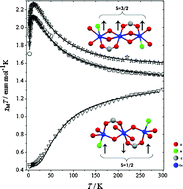
The linear trinuclear Cu(II) complexes [Cu3(L1)4(H2tea)2] (1), [Cu3(L2)4(H2tea)2]·2CH3CN (2), [Cu3(L2)4(H2tea)2] (3), [Cu3(L1)2(H2tea)2(NO3)2] (4) and the dinuclear complex [Cu2(L1)2(H2tea)2] (5), where L1 = 2-thiophene carboxylate, L2 = 2-(thiophen-2-yl)-acetate and H2tea = the single deprotonated form of triethanolamine have been prepared and characterised while the crystal structures of 1–4 have been determined. The variable-temperature magnetic susceptibilities of complexes 1–5 have been measured in the range 2–300 K under various external fields in the range 0.02–1.0 T. X-band EPR spectra of 1–5 compounds were recorded at 4–100 K. Complexes 1, 2 and 3 found to have the same J = 33 cm−1 and g values 2.16(1), 2.20(1) and 2.16(1) respectively while for 5J = 15 cm−1 and g = 2.06(1) revealing a clear ferromagnetic exchange between Cu(II) ions. Complex 4 was found to be antiferromagnetic with J = −28 cm−1 and g = 2.21(1). The polycrystalline powder X-band EPR spectrum of complexes 1, 2, and 3 at 4 K are dominated by a transition at 1600 G (g = 4.3) which unambiguously identifies the spin of the ground multiplet (S = 3/2) while the antiferromagnetic complex 4 has a derivative centered at g = 2.1 indicative of a ground doublet (S = 1/2). Concerning complex 5 a spectrum of a dominant derivative centered at g = 2.06(1) is observed with a very weak half field transition (ca. 1500 G) indicative of the ferromagnetic nature of the system. Furthermore, for complexes 2 and 3 a strong temperature dependence of this spectroscopic g-factor is revealed and change of the geff from the liquid helium temperature to the room temperature is almost 2 units.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...