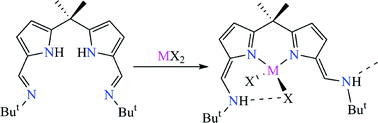
The synthesis and structures of Fe, Co, and Zn halide complexes [MX2(H2L)] (M = Fe, X = Br; M = Co, Zn, X = Cl) of the N-donor extended dipyrromethane ligand H2L are described, from which it is clear that bond rearrangements from imine-pyrrole to amine-azafulvene tautomers occur on metal co-ordination, both in the solid state and in solution. In the structure of [FeBr2(H2L)], this H-migration results in a pendant amine that is involved in both inter- and intramolecular hydrogen bonds to the bromide ligands, so forming a dimer. As the tautomerisation renders the N–H protons less acidic, metal-based ligand substitution reactions can occur in favour of deprotonation. As such, the reaction between [MCl2(H2L)] (M = Co, Zn) and NaN3 results in the formation of the bis(azide) complexes [M(N3)2(H2L)] which for Co displays both inter- and intramolecular N–H⋯N3–Co hydrogen bonds in the solid state. In contrast, reactions of the dihalides with the lithium bases LiNMe2 or LiMe (M = Fe), or reduction reactions with C8K (M = Fe, Co) result in the formation of the known dinuclear helicates [M2(L)2].
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...