A comparative analysis of the functionalisation of unactivated cycloalkanes using alkynes and either sunlight or a photochemical reactor
Abstract
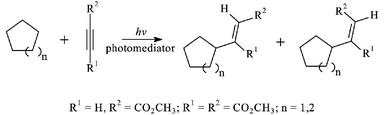
Using a standard mercury vapour lamp or sunlight, the synthetically difficult task of introducing functionality into unactivated

 Please wait while we load your content...
Please wait while we load your content...