Glycosylation of a model proto-RNA nucleobase with non-ribose sugars: implications for the prebiotic synthesis of nucleosides†
Abstract
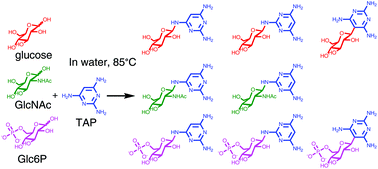
The emergence of nucleosides is an important, but poorly understood, element of the origins of life. We show that 2,4,6-triaminopyrimidine (TAP), a possible ancestral nucleobase of RNA, is glycosylated in water by non-ribose sugars in yields comparable to those previously reported for its reaction with ribose. The various sugars surveyed include ketoses and aldoses; tetroses, pentoses, and hexoses and are neutral, anionic, or cationic. Though they vary greatly in structure and properties, the data show that all sugars tested form glycosides with TAP. The structures of the eight TAP glycosides formed with glucose and two of its derivatives, glucose-6-phosphate and N-acetylglucosamine, were found to be β-pyranosides with the glycosylation site on TAP varying with sugar identity. Our results suggest that prebiotic nucleoside formation would not have been restricted to ribose if ancestral RNA (or proto-RNA) utilized TAP and/or other proto-nucleobases with similar reactivities, and that the ability to form higher-order structures may have influenced proto-RNA monomer selection.
- This article is part of the themed collection: Nucleic Acid Modifications


 Please wait while we load your content...
Please wait while we load your content...