A simple and novel protocol for Li-trapping with a POM/MOF nano-composite as a new adsorbent for CO2 uptake†
Abstract
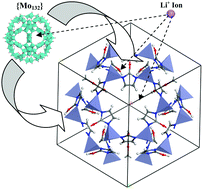
In the present article, a convenient and novel procedure for lithium ion-trapping over a polyoxometalate/metal organic framework nano-composite to synthesize a new class of porous composite materials for carbon capturing has been demonstrated. For this purpose, the successful interaction of zeolitic imidazolate framework-8 (ZIF-8) with a water-soluble Keplerate-type polyoxometalate ({Mo132}) is accomplished and a POM/ZIF-8 nano-composite material (ZIF-8@{Mo132}) is achieved. The POM/ZIF-8 nano-composite enables the uptake of lithium functional groups (Li+ cations) due to the existence of {Mo132} in its structure. As a result of interactions between ZIF-8@{Mo132} and an organolithium compound, a Li-trapped POM/ZIF-8 nano-composite material (ZIF-8@{Mo132}@Li+) is readily obtained. In particular, the ZIF-8@{Mo132}@Li+ nano-composite displays a surprising enhancement in CO2 adsorption capacity, the most significant greenhouse gas. The CO2 uptake of ZIF-8@{Mo132}@Li+ is around 26.40 wt%, while the capacity of pristine ZIF-8 is only around 4 wt%. The presence of lithium cations inside and on the surface of the as-synthesized nano-composite plays a key role in the enhancement of CO2 storage.


 Please wait while we load your content...
Please wait while we load your content...