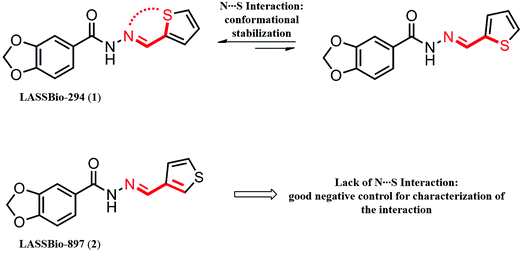
Theoretical and experimental characterization of 1,4-N⋯S σ-hole intramolecular interactions in bioactive N-acylhydrazone derivatives†
Pedro de Sena Murteira
Pinheiro
ab,
Daniel Alencar
Rodrigues
ac,
Marina Amaral
Alves
ac,
Luzineide Wanderley
Tinoco
 bd,
Glaucio Braga
Ferreira
e,
Carlos Mauricio Rabello
de Sant’Anna
af and
Carlos Alberto Manssour
Fraga
bd,
Glaucio Braga
Ferreira
e,
Carlos Mauricio Rabello
de Sant’Anna
af and
Carlos Alberto Manssour
Fraga
 *ab
*ab
aLaboratório de Avaliação e Síntese de Substâncias Bioativas (LASSBio), Instituto de Ciências Biomédicas, Universidade Federal do Rio de Janeiro, PO Box 68023, 21941-902, Rio de Janeiro, RJ, Brazil. E-mail: cmfraga@ccsdecania.ufrj.br
bPrograma de Pós-Graduação em Farmacologia e Química Medicinal, Instituto de Ciências Biomédicas, Universidade Federal do Rio de Janeiro, 21941-902, Rio de Janeiro, RJ, Brazil
cPrograma de Pós-Graduação em Química, Instituto de Química, Universidade Federal do Rio de Janeiro, 21941-909, Rio de Janeiro, RJ, Brazil
dLaboratório de Análise e Desenvolvimento de Inibidores Enzimáticos (LADIE), Instituto de Pesquisa em Produtos Naturais (IPPN), Universidade Federal do Rio de Janeiro, 21941-902, Rio de Janeiro, RJ, Brazil
ePrograma de Pós-Graduação em Química, Instituto de Química, Universidade Federal Fluminense, Niterói, Rio de Janeiro, 24020-141, Brazil
fDepartamento de Química, Instituto de Ciências Exatas, Universidade Federal Rural do Rio de Janeiro, 23970-000, Seropédica, RJ, Brazil
First published on 28th November 2017
Abstract
Sigma-hole (σ-hole) bonds are interactions that are gaining special attention in medicinal chemistry. This type of interaction, initially assigned to the halogens (group 17 of the periodic table), has been extended to atoms of groups 14, 15 and 16. Sulfur atoms have been outstanding for describing these interactions at the intramolecular level (to induce conformational stability) and the intermolecular level (participating in molecular recognition of bioactive compounds by their respective targets). Thus, this work describes the theoretical and experimental characterization of a 1,4-N⋯S σ-hole intramolecular interaction in the N-acylhydrazone cardioactive prototype LASSBio-294 (1), which leads to conformational stabilization and has a direct influence on the molecular properties of this inotropic prototype compared to a negative control for the interaction, LASSBio-897 (2), which is the regioisomer at the thiophene ring. Our theoretical results were reached using the B3LYP/6-311G(d) level of theory, including analysis of conformational, orbital and electrostatic properties. We performed experimental studies using IR, Raman, UV and NMR spectroscopies, which corroborated our theoretical data, showing significant differences between LASSBio-294 (1) and LASSBio-897 (2) in relation to the bond strength of the groups involved in the N⋯S interaction (S–C and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds), the energies of the orbitals associated with the S lone pair (Lp(S)) and the antibonding N
C bonds), the energies of the orbitals associated with the S lone pair (Lp(S)) and the antibonding N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C π bond (π*(N
C π bond (π*(N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)), as well as the 15N chemical shifts in both systems. Together, our results show how this unusual interaction can influence the molecular properties of some organic compounds.
C)), as well as the 15N chemical shifts in both systems. Together, our results show how this unusual interaction can influence the molecular properties of some organic compounds.
1. Introduction
Sigma-hole interactions were initially associated with halogens – specifically chlorine, bromine and iodine – which demonstrated their ability to interact with Lewis bases (nitrogen, oxygen, sulfur and π systems)1,2 through regions of positive electrostatic potential located on their surfaces (σ-hole regions). These regions, originating from low-lying σ* orbitals of the C–X bond (X = Cl, Br, I), allow favorable interactions with available electron-donating groups.3,4The importance of the σ-hole interactions involving elements of group 17, also called halogen bonds, has been recognized and exploited in the rational design of drug candidates.3,5,6 Some interesting examples of this interaction are involved in the molecular recognition of bioactive compounds by their respective biotargets.5,7–9
Other atoms are also attracting interest for their σ-hole interactions, including sulfur, where σ-hole regions are present on both sides of the atom (opposite the C–S bonds) allowing both intramolecular and intermolecular interactions with Lewis bases.10,11 As can be observed in the work published by Burling and Goldstein,12 who performed an investigation in the Cambridge Structural Database (CSD) and identified 78 structures containing a 1,4-N⋯S relationship, among which 69% showed the two atoms separated by a distance smaller than the sum of their van der Waals radii.
These interactions, mediated by sulfur atoms, were recently reviewed by Beno and co-workers (2015),3 highlighting that these σ-hole intramolecular interactions can induce conformational stabilization of compounds, which can consequently influence the interaction mode of bioactive compounds with their respective targets.13–15
Murray and colleagues (2008)16 studied this interaction, and these researchers analyzed the influence of electron-withdrawing and electron-donating groups in the sulfur σ-hole regions theoretically. In their report, electron withdrawing groups linked to a thiazole ring, such as –CN and –Cl, increased the electrostatic potential of the σ-hole regions; presence of electron-donating groups, such as –NH2, decreased the electrostatic potential of these regions, promoting a direct effect on the interaction energy between the sulfur atoms and Lewis bases.
Similar reports describing interactions with other groups that can act as Lewis bases were also published. Markham and Bock (1997) carried out theoretical research on 1,4-O⋯S intramolecular interactions and found that these interactions are very sensitive to the electronegative character of the group linked to the sulfur atom.17 Also related to the O⋯S intramolecular interactions, Rittner and colleagues (2011) performed an investigative study on the conformational preferences of 5-substituted 2-acetyl-thiophenes using theoretical calculations and infrared spectroscopy.18 They determined a population of approximately 80% for the O,S-cis conformation, highlighting the importance of this type of interaction in fixing specific conformations.18 Moreover, Zhang and colleagues (2015) reported the characterization of O⋯S intermolecular interactions through the use of theoretical calculations and statistical analysis in derivatives presenting thiophene, thiazole and thiadiazole rings.11 They showed increased stability of the O⋯S intermolecular interaction when the sulfur atom is present in thiazole and thiadiazole rings, which may be a consequence of increasing both the electrostatic potential and the size of the σ-hole regions.11
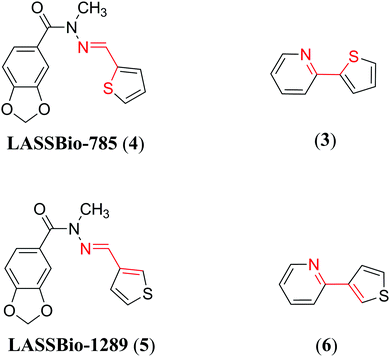
Thus, the importance of σ-hole interactions involving sulfur atoms is just now coming to light. Our group is interested in these interactions in bioactive compounds such as the cardioactive compound LASSBio-294 (1) (Fig. 1).19 This N-acylhydrazone derivative was developed by our research group and shows potent inotropic effects in isolated hearts of Wistar rats20 and moderate vasodilator activity in the intact aortic rings of Wistar rats.21 It is also able to prevent exercise intolerance and cardiac remodeling in rats submitted to myocardial infarction.22,23 These data make this compound an interesting drug candidate for the treatment of chronic heart failure. Many efforts were performed to optimize LASSBio-294 (1), but none were successful. One of the most interesting examples confirms that optimization of LASSBio-294 (1) is difficult, namely, the analysis of its regioisomer at the thiophene ring, LASSBio-897 (2) (Fig. 1). LASSBio-897 (2) showed an improved vasodilatory activity, but no inotropic effect.24 Based on recent publications3,14,25,26 and data about LASSBio-294 (1) and LASSBio-897 (2), we believe that the 1,4-N⋯S relationship present in the structure of LASSBio-294 (1) is important for a N⋯S σ-hole intramolecular interaction and possibly for the range of pharmacological activities evidenced for this cardioactive prototype. Beyond that, N-acylhydrazones are known privileged structures that can be used in the pharmacological modulation of several biological targets.27–29
Thus, in this work, we investigated, for the first time, the possibility of a N⋯S σ-hole intramolecular interaction in the N-acylhydrazone derivative LASSBio-294 (1), through the combined use of molecular modeling and spectroscopic techniques.
2. Methods
2.1. Molecular modeling
Potential energy surface (PES) scan analysis and equilibrium geometry calculations were performed with Spartan'16 software (Wavefunction, Inc.). NBO (Natural Bond Orbitals) analysis30 and spectra calculations were performed using the Gaussian'09 software package.31 Theoretical calculations were performed with the B3LYP32,33 density functional level of theory using the 6-311G(d) basis set. Vibrational spectra calculations in the gas phase and UV spectra in DMSO were calculated using PCM solvent model34,35 with the TD-SCF excited state method for comparison with experimental results. Additionally, the total energy distribution analysis (TED) of the theoretical vibrational spectra was obtained using VEDA436 software, which allowed us to pinpoint the vibrational modes related to each frequency in the experimental and theoretical spectra.2.2. Chemistry
LASSBio-294 (1) and LASSBio-897 (2) were synthesized according to the proceedings described by Kümmerle et al. (2009).40 Characterization data for LASSBio-294 (1) and LASSBio-897 (2) are described in the ESI,† Section 4 (Fig. S14 and S15).2.3. Spectroscopic analysis
We conducted a spectroscopic analysis of the compounds LASSBio-294 (1) and LASSBio-897 (2) to experimentally evaluate the 1,4-N⋯S σ-hole intramolecular interaction. LASSBio-294 (1) was used as a positive control for the interaction and LASSBio-897 (2) was used as a negative control. These compounds were evaluated through NMR, IR, Raman and UV spectroscopy, the acquisition details can be found in the ESI† (Sections 5, 5.1–5.3).3. Results and discussion
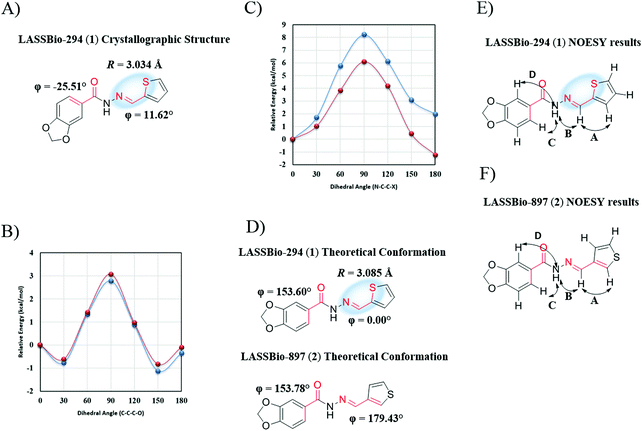
An initial indication of the occurrence of 1,4-N⋯S σ-hole intramolecular interactions in the cardioactive compound LASSBio-294 (1) can be observed through analysis of its crystallographic structure,40 which showed a distance between the nitrogen and sulfur atoms of 3.034 Å (smaller than the sum of their van der Waals radii) associated with a dihedral angle (N–C–C–S) of 11.62° (Fig. 2A).3We first performed theoretical calculations to evaluate the intramolecular interaction and then performed experimental evaluations to validate the theoretical data. At all stages, we used LASSBio-897 (2) as a negative control for this intramolecular interaction, due to the great structural similarity between both compounds; the sulfur position in the thiophene ring of LASSBio-897 (2) makes the N⋯S interaction impossible.
3.1. Conformational analysis
The theoretical evaluation of the PES scan with B3LYP/6-311G(d) through the dihedral angle related to the conformation of the benzodioxole in relation to the carbonyl of LASSBio-294 (1) demonstrated an inversion of ∼180° for the lowest energy conformer compared to the crystallographic structure (Fig. 2A and B). We used the lowest energy conformer observed in this previous analysis for the subsequent evaluation of the dihedral angle related to the 1,4-N⋯S σ-hole intramolecular interaction, which lead to a difference of 2 kcal mol−1 between the syn (ϕ = 0°) conformer and the anti conformer (ϕ = 180°) (Fig. 2C). This highlights the greater stability of the syn conformer, in agreement with other theoretical data from the literature.3,25,41 In addition, analysis of the Boltzmann conformational distribution at 298.15 K indicated 91.2% of the syn conformer for LASSBio-294 (1). The PES scan related to the conformation of the benzodioxole in relation to the carbonyl for LASSBio-897 (2) was similar to that obtained for LASSBio-294 (1) (Fig. 2B). In relation to the conformation of the thiophene ring, however, it was possible to observe that the anti conformer was favored by 1.2 kcal mol−1, which represents 82.5% of the conformers according to the Boltzmann distribution at 298.15 K (Fig. 2C). The Cartesian coordinates of the final geometry of LASSBio-294 (1) and LASSBio-897 (2) are available in the ESI,† Section 6 (Table S18).To validate the conformational theoretical data, we conducted NOESY-2D analyses, where it was possible to observe spatial interactions between the hydrogens in LASSBio-294 (1) (A = δ 8.62, 7.43; B = δ 11.63, 8.62; C = δ 11.63, 7.47; D = δ 11.63, 7.43) and LASSBio-897 (2) (A = δ 8.50, 7.51; B = δ 11.61, 8.50; C = δ 11.61, 7.53; D = δ 11.63, 7.44), which corroborated our theoretical data (Fig. 2E and F). The interaction A in LASSBio-294 (1) is related to the conformation of the thiophene ring in relation to the imine moiety, which demonstrated the stability of the syn conformer, as predicted by the PES scan analysis. This NOESY-2D analysis showed the proximity of the nitrogen and sulfur atoms in DMSO for LASSBio-294 (1), that is another indication of the presence of a σ-hole 1,4-N⋯S intramolecular interaction. In addition, it was possible to identify two conformers in relation to the benzodioxole for LASSBio-294 (1) and LASSBio-897 (2) (Fig. 2E and F), which according to our theoretical data, it is probably due to the low energy rotational barrier (∼3 kcal mol−1) and to the small energy difference between these two conformers (∼0.3 kcal mol−1) (Fig. 2B).
3.2. Orbital and electronic analysis
To obtain more specific data about the intramolecular interaction, other theoretical and experimental analyses were performed. From the theoretical evaluation with B3LYP/6-311G(d), bond order, charges (Mulliken, NBO and CHELPG42) and NBO data were obtained and were corroborated by spectroscopic analysis.σ-hole interactions are described in the literature both (a) from an electrostatic point of view, using Coulomb's Law,2,43 where the nitrogen atom carries a partial negative charge and the sulfur atom carries a partial positive charge; and (b) from the orbital point of view, as an interaction between an isolated electron pair, for example the electron lone pair of an imine nitrogen, and one of the σ* orbitals of the sulfur–carbon bonds.3,4,18
According to the Mulliken, NBO and CHELPG charges (Table 1), the sulfur atom can behave as an electrophile in both LASSBio-294 (1) and LASSBio-897 (2), with a more positive partial charge in LASSBio-294 (1) according to the Mulliken and NBO charges, which is a consequence of the greater delocalization of the perpendicular sulfur lone pair through the thiophene ring. The charge data also showed that the imine nitrogen can act as a nucleophile in both systems. However, the Mulliken charges showed that the imine nitrogen of LASSBio-294 (1) is more nucleophilic than LASSBio-897 (2), while the NBO charges indicated that the imine nitrogen has the same charge in both systems and the CHELPG charges indicated that the imine nitrogen of LASSBio-294 (1) is less nucleophilic than LASSBio-897 (2). According to the 15N chemical shift values detected by (1H–15N) HMBC experiments the imine nitrogen of LASSBio-294 (1) is more shielded than in LASSBio-897 (2) (Table 1), which is only corroborated by the Mulliken charges. In addition, chemical shift values indicated that the amide nitrogen atoms present the same charge in both systems, which was confirmed by the Mulliken's charge calculations, but it was not confirmed by NBO and CHELPG charges (Table 1). In general, regardless of the type of charge used, the theoretical data indicated that in LASSBio-294 (1) the imine nitrogen atom has a negative partial charge and the sulfur atom has a positive partial charge, which reinforces the electrostatic point of view of the interaction between these two atoms. This is also corroborated by the chemical shielding effect of the imine nitrogen of LASSBio-294 (1) in relation to LASSBio-897 (2), since this effect is similar to other types of interactions, such as intramolecular hydrogen bonds, which has a strong electrostatic character.44,45 These data further corroborate our theoretical and experimental observations.
| Imine 15N chemical shift (ppm) | −67.4 | −63.0 |
| Amide 15N chemical shift (ppm) | −212.2 | −212.0 |
| Imine N Mulliken charges | −0.134 | −0.126 |
| Imine N NBO charges | −0.227 | −0.227 |
| Imine N CHELPG charges | −0.200 | −0.224 |
| Amide N Mulliken charges | −0.492 | −0.492 |
| Amide N NBO charges | −0.421 | −0.423 |
| Amide N CHELPG charges | −0.289 | −0.237 |
| S Mulliken charges | 0.326 | 0.300 |
| S NBO charges | 0.444 | 0.405 |
| S CHELPG charges | 0.023 | 0.037 |
From an orbital point of view, the Mulliken bond order analysis showed a direct interaction between the nitrogen and sulfur atoms (0.060) for LASSBio-294 (1), indicative of a weak interaction between these atoms. In addition, it was possible to identify an electronic transition area through the analysis of the electrostatic potential map of LASSBio-294 (1) (Fig. 3). We also performed NBO analysis to observe interactions between the Lp(N) orbital and the σ*(S–C) (Lp(N) → σ*(S–C)) in LASSBio-294 (1). However, an interaction between these orbitals was not observed, suggesting that in LASSBio-294 (1) this interaction energy is lower than 0.5 kcal mol−1. It is expected that in LASSBio-294 (1) the interaction has more electrostatic character, possibly because of the 1,4-N⋯S relationship, which prevents effective orbital overlap.3
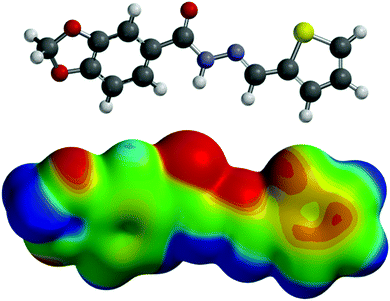 | ||
| Fig. 3 Molecular electrostatic potential map of LASSBio-294 (1) (B3LYP/6-311G(d)) at the 0.002 a.u. contour level. The map are in the range of +100 to −100 kJ mol−1. | ||
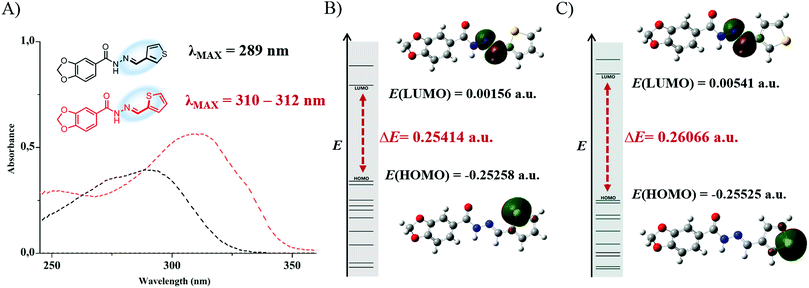
We also observed the orbital energy differences of the HOMO, classified as sulfur lone pair – Lp(S), and the LUMO, the π*(N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) orbital in both systems, through the UV spectra calculations and NBO analysis. In LASSBio-294 (1), an increase in the Lp(S) orbital energy was observed compared to the Lp(S) orbital of LASSBio-897 (2) (Fig. 4B and C), which was probably due to the field effect caused by the spatial approximation of the imine nitrogen atom.
C) orbital in both systems, through the UV spectra calculations and NBO analysis. In LASSBio-294 (1), an increase in the Lp(S) orbital energy was observed compared to the Lp(S) orbital of LASSBio-897 (2) (Fig. 4B and C), which was probably due to the field effect caused by the spatial approximation of the imine nitrogen atom.
In addition, due to the increased energy of the HOMO (Lp(S)) orbital in LASSBio-294 (1), there is greater delocalization of this electron pair toward the thiophene ring, as can be seen in Table 2, in which the Lp(S) orbital of LASSBio-294 (1) interacts with the π* adjacent orbitals of the thiophene ring with ∼1.8 kcal mol−1 higher in relation to LASSBio-897 (2). Consequently, there is also a stronger resonance between the imine moiety and the thiophene ring of LASSBio-294 (1) (∼2 kcal mol−1 higher) in comparison to LASSBio-897 (2) (Table 2). With this stronger resonance, it would be expected a lower energy of the LUMO orbital for LASSBio-294 (1) in relation to LASSBio-897 (2), due to the greater electron donation to the π*(N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) orbital, which was also corroborated by the theoretical data (Fig. 4B and C). Consequently, there is a lower energy gap between the HOMO and LUMO orbitals in LASSBio-294 (1) in comparison to LASSBio-897 (2), being expected a longer λMAX absorption in the UV spectra analysis for LASSBio-294 (1). These theoretical analysis were corroborated by our experimental data, where a bathochromic shift of the λMAX of LASSBio-294 (1) was observed in relation to the λMAX of LASSBio-897 (2), highlighting the ΔE(HOMO–LUMO) difference of both systems (Fig. 4A). Our theoretical UV spectra calculations confirmed that the λMAX absorption bands are related to HOMO–LUMO transitions in both LASSBio-294 (1) (oscillator strength = 0.8242) and LASSBio-897 (2) (oscillator strength = 0.8146) (see ESI,† Section 4 and Table S17).
C) orbital, which was also corroborated by the theoretical data (Fig. 4B and C). Consequently, there is a lower energy gap between the HOMO and LUMO orbitals in LASSBio-294 (1) in comparison to LASSBio-897 (2), being expected a longer λMAX absorption in the UV spectra analysis for LASSBio-294 (1). These theoretical analysis were corroborated by our experimental data, where a bathochromic shift of the λMAX of LASSBio-294 (1) was observed in relation to the λMAX of LASSBio-897 (2), highlighting the ΔE(HOMO–LUMO) difference of both systems (Fig. 4A). Our theoretical UV spectra calculations confirmed that the λMAX absorption bands are related to HOMO–LUMO transitions in both LASSBio-294 (1) (oscillator strength = 0.8242) and LASSBio-897 (2) (oscillator strength = 0.8146) (see ESI,† Section 4 and Table S17).
Although our theoretical analysis indicated that the 1,4-N⋯S σ-hole intramolecular interaction in LASSBio-294 (1) has higher electrostatic character, it was possible to experimentally observe the presence of a relevant orbital character in this interaction using IR and Raman spectroscopic analysis. Using theoretical spectra calculations, it was possible to perform a total energy distribution analysis36 to determine the main bands related to the vibrations of the S–C and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds (see ESI,† Section 4, Tables S15 and S16).46 Significant differences were observed in the main stretching wavenumbers of the S–C and N
C bonds (see ESI,† Section 4, Tables S15 and S16).46 Significant differences were observed in the main stretching wavenumbers of the S–C and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds in LASSBio-294 (1) and LASSBio-897 (2) (Fig. 5). The S–C stretching wavenumbers in LASSBio-294 (1) appeared at 758 cm−1, whereas in LASSBio-897 (2) it appeared at 848 cm−1. In relation to the imine bonds (N
C bonds in LASSBio-294 (1) and LASSBio-897 (2) (Fig. 5). The S–C stretching wavenumbers in LASSBio-294 (1) appeared at 758 cm−1, whereas in LASSBio-897 (2) it appeared at 848 cm−1. In relation to the imine bonds (N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) it is possible to observe that in LASSBio-294 (1), the stretching wavenumber appeared at 1592 cm−1 while in LASSBio-897 (2) it appeared at 1606 cm−1, in agreement with the observations of NBO analysis for the resonance between the thiophene ring and the imine moiety of the N-acylhydrazone subunit (Table 2). The lower stretching wavenumber of the N
C) it is possible to observe that in LASSBio-294 (1), the stretching wavenumber appeared at 1592 cm−1 while in LASSBio-897 (2) it appeared at 1606 cm−1, in agreement with the observations of NBO analysis for the resonance between the thiophene ring and the imine moiety of the N-acylhydrazone subunit (Table 2). The lower stretching wavenumber of the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in LASSBio-294 (1) is experimentally indicative of the stronger resonance, as there is higher electronic donation to the π*(N
C bond in LASSBio-294 (1) is experimentally indicative of the stronger resonance, as there is higher electronic donation to the π*(N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) orbital (∼3.3 kcal mol−1, Table 2), decreasing the bond strength of the imine bond in LASSBio-294 (1) in comparison to LASSBio-897 (2).
C) orbital (∼3.3 kcal mol−1, Table 2), decreasing the bond strength of the imine bond in LASSBio-294 (1) in comparison to LASSBio-897 (2).
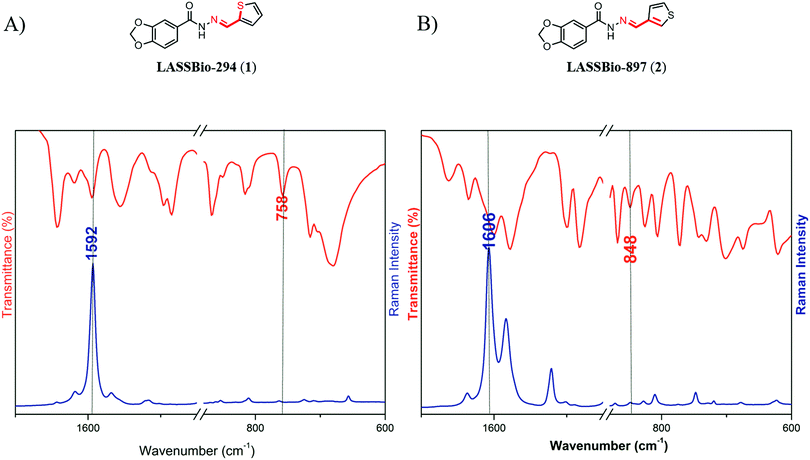 | ||
| Fig. 5 Data from IR (red) and Raman (blue) spectroscopy in the region of 1700–1450/880–600 cm−1. (A) Data obtained for LASSBio-294 (1). (B) Data obtained for LASSBio-897 (2). | ||
Taking into account the theoretical and experimental data presented in this work, it is possible to confirm that there is an N⋯S intramolecular interaction in the N-acylhydrazone derivative LASSBio-294 (1), which causes a conformational restriction and has relevant consequences in its molecular properties when compared to its regioisomer, LASSBio-897 (2). This observation has implications in the medicinal chemistry community, since this type of interaction is only beginning to be understood and there are some other interesting examples in which N-acylhydrazone derivatives substituted with a 2-thiophene ring at the imine side were used and presented several pharmacological activities, such as antiviral,47 antibacterial,48,49 analgesic and anti-inflammatory.50 Beyond that, the study of the molecular properties of N-acylhydrazone derivatives associated with the N⋯S intramolecular interaction can help in the understanding of the photoswitch properties of this privileged structure.51 To our knowledge, this is the first report of a N⋯S interaction involving the privileged structure N-acylhydrazone and considering that this molecular fragment has been extensively explored in the pharmacological modulation of several biological targets,28,29,52 we hope that this work will open new doors for the rational use of this privileged structure, such as exploring conformationally restricted compounds through a N⋯S σ-hole interaction.3,13–15
To explore the analysis performed here, we applied the same studies in two similar systems with N-methylation of the amide nitrogen of LASSBio-294 (1) and LASSBio-897 (2), i.e., LASSBio-785 (4)40,53,54 and LASSBio-1289 (5),55–57 respectively (Fig. 6). The N-methylation of these compounds causes significant conformational changes,40,58 but did not affect the occurrence of the 1,4-N⋯S σ-hole intramolecular interaction in LASSBio-785 (4), as significant differences were also observed in the bond strength of the S–C and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds, including a bathochromic shift of the λMAX in the UV spectra and the 15N chemical shifts in relation to LASSBio-1289 (5) (see ESI,† Section 3, Fig. S9–S13 and Tables S12–S14). These analyses were applied in two other systems, a positive control of these interactions already established in the literature 2-(thiophen-2-yl)pyridine (3)37,41 (used in our theoretical validation) and its respective negative control 2-(thiophen-3-yl)pyridine (6) (Fig. 6), which validated the results for the N-acylhydrazone derivatives (see ESI,† Section 2, Fig. S4–S8 and Tables S9–S11).
C bonds, including a bathochromic shift of the λMAX in the UV spectra and the 15N chemical shifts in relation to LASSBio-1289 (5) (see ESI,† Section 3, Fig. S9–S13 and Tables S12–S14). These analyses were applied in two other systems, a positive control of these interactions already established in the literature 2-(thiophen-2-yl)pyridine (3)37,41 (used in our theoretical validation) and its respective negative control 2-(thiophen-3-yl)pyridine (6) (Fig. 6), which validated the results for the N-acylhydrazone derivatives (see ESI,† Section 2, Fig. S4–S8 and Tables S9–S11).
4. Conclusion
In this work, we examined the 1,4-N⋯S σ-hole intramolecular interaction in LASSBio-294 (1) using theoretical and experimental evaluations and found that this interaction modifies the molecular properties of LASSBio-294 (1) significantly in relation to its regioisomer, LASSBio-897 (2), which played a key role in our analysis by acting as a negative control of these interactions. Our experimental data, achieved through NMR, UV, IR and Raman spectroscopy, supports our theoretical results and strongly indicates the occurrence of an intramolecular interaction between the nitrogen and sulfur atoms in LASSBio-294 (1), with higher electrostatic character.To our knowledge, this is the first time that σ-hole interactions have been investigated simultaneously using different spectroscopic techniques and theoretical evaluations and a negative control, which strengthens our results. This is also the first description of N⋯S σ-hole intramolecular interactions in N-acylhydrazone derivatives, which is very important because this versatile privileged structure can be used in the modulation of several biological targets. We hope that our results will assist with further characterizations of this type of interaction.
Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Funding sources
This work was funded by CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), INCT-Inofar (Instituto Nacional de Ciência e Tecnologia de Fármacos e Medicamentos, BR, Grant No. 573.564/2008-6) and FAPERJ (Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro).Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors would like to thank the multi-user analysis laboratories LAMAR (Laboratório Multiusuário de Análises por RMN – Instituto de Pesquisas de Produtos Naturais, UFRJ) and LAME (Laboratório Multiusuário de Espectroscopia – UFF) for the realization of the NMR and IR and Raman experiments, respectively.References
- Y. Lu, T. Shi, Y. Wang, H. Yang, X. Yan, X. Luo, H. Jiang and W. Zhu, J. Med. Chem., 2009, 52, 2854–2862 CrossRef CAS PubMed
.
- P. Politzer, J. S. Murray and T. Clark, Phys. Chem. Chem. Phys., 2010, 12, 7748 RSC
.
- B. R. Beno, K.-S. Yeung, M. D. Bartberger, L. D. Pennington and N. A. Meanwell, J. Med. Chem., 2015, 58, 4383–4438 CrossRef CAS PubMed
.
- F. Zhou, R. Liu, J. Tang, P. Li, Y. Cui and H. Zhang, J. Mol. Model., 2016, 22, 29 CrossRef PubMed
.
- R. Wilcken, M. O. Zimmermann, A. Lange, A. C. Joerger and F. M. Boeckler, J. Med. Chem., 2013, 56, 1363–1388 CrossRef CAS PubMed
.
- S. Sirimulla, J. B. Bailey, R. Vegesna and M. Narayan, J. Chem. Inf. Model., 2013, 53, 2781–2791 CrossRef CAS PubMed
.
- P. Auffinger, F. A. Hays, E. Westhof and P. S. Ho, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 16789–16794 CrossRef CAS PubMed
.
- M. R. Scholfield, C. M. Vander Zanden, M. Carter and P. S. Ho, Protein Sci., 2013, 22, 139–152 CrossRef CAS PubMed
.
- A. Lange, M. Günther, F. M. Büttner, M. O. Zimmermann, J. Heidrich, S. Hennig, S. Zahn, C. Schall, A. Sievers-Engler, F. Ansideri, P. Koch, M. Laemmerhofer, T. Stehle, S. A. Laufer and F. M. Boeckler, J. Am. Chem. Soc., 2015, 137, 14640–14652 CrossRef CAS PubMed
.
- A. L. Ringer, A. Senenko and C. D. Sherrill, Protein Sci., 2007, 16, 2216–2223 CrossRef CAS PubMed
.
- X. Zhang, Z. Gong, J. Li and T. Lu, J. Chem. Inf. Model., 2015, 55, 2138–2153 CrossRef CAS PubMed
.
- F. T. Burling and B. M. Goldstein, Acta Crystallogr., Sect. B: Struct. Sci., 1993, 49(Pt 4), 738–744 CrossRef
.
- J. Hynes, H. Wu, S. Pitt, D. R. Shen, R. Zhang, G. L. Schieven, K. M. Gillooly, D. J. Shuster, T. L. Taylor, X. Yang, K. W. McIntyre, M. McKinnon, H. Zhang, P. H. Marathe, A. M. Doweyko, K. Kish, S. E. Kiefer, J. S. Sack, J. A. Newitt, J. C. Barrish, J. Dodd and K. Leftheris, Bioorg. Med. Chem. Lett., 2008, 18, 1762–1767 CrossRef CAS PubMed
.
- S. Lin, S. T. Wrobleski, J. Hynes, S. Pitt, R. Zhang, Y. Fan, A. M. Doweyko, K. F. Kish, J. S. Sack, M. F. Malley, S. E. Kiefer, J. A. Newitt, M. McKinnon, J. Trzaskos, J. C. Barrish, J. H. Dodd, G. L. Schieven and K. Leftheris, Bioorg. Med. Chem. Lett., 2010, 20, 5864–5868 CrossRef CAS PubMed
.
- L. D. Pennington, M. D. Bartberger, M. D. Croghan, K. L. Andrews, K. S. Ashton, M. P. Bourbeau, J. Chen, S. Chmait, R. Cupples, C. Fotsch, J. Helmering, F.-T. Hong, R. W. Hungate, S. R. Jordan, K. Kong, L. Liu, K. Michelsen, C. Moyer, N. Nishimura, M. H. Norman, A. Reichelt, A. C. Siegmund, G. Sivits, S. Tadesse, C. M. Tegley, G. Van, K. C. Yang, G. Yao, J. Zhang, D. J. Lloyd, C. Hale and D. J. St. Jean, J. Med. Chem., 2015, 58, 9663–9679 CrossRef CAS PubMed
.
- J. S. Murray, P. Lane and P. Politzer, Int. J. Quantum Chem., 2008, 108, 2770–2781 CrossRef CAS
.
- G. D. Markham and C. W. Bock, THEOCHEM, 1997, 418, 139–154 CrossRef CAS
.
- R. Rittner, L. C. Ducati, C. F. Tormena, B. C. Fiorin and C. B. Braga, Spectrochim. Acta, Part A, 2011, 79, 1071–1076 CrossRef CAS PubMed
.
- P. C. Lima, L. M. Lima, K. C. M. da Silva, P. H. O. Léda, A. L. P. de Miranda, C. A. M. Fraga and E. J. Barreiro, Eur. J. Med. Chem., 2000, 35, 187–203 CrossRef CAS PubMed
.
- R. T. Sudo, G. Zapata-Sudo and E. J. Barreiro, Br. J. Pharmacol., 2001, 134, 603–613 CrossRef CAS PubMed
.
- C. L. M. Silva, F. Noël and E. J. Barreiro, Br. J. Pharmacol., 2002, 135, 293–298 CrossRef CAS PubMed
.
- J. S. da Silva, S. L. Pereira, R. do, C. Maia, S. S. Landgraf, C. Caruso-Neves, A. E. Kümmerle, C. A. M. Fraga, E. J. Barreiro, R. T. Sudo and G. Zapata-Sudo, Life Sci., 2014, 94, 30–36 CrossRef PubMed
.
- D. G. Costa, J. S. da Silva, A. E. Kümmerle, R. T. Sudo, S. S. Landgraf, C. Caruso-Neves, C. A. M. Fraga, E. J. de Lacerda Barreiro and G. Zapata-Sudo, Am. J. Hypertens., 2010, 23, 1220–1227 CrossRef CAS PubMed
.
- G. Zapata-Sudo, S. L. Pereira, H. J. V. Beiral, A. E. Kummerle, J. M. Raimundo, F. Antunes, R. T. Sudo, E. J. Barreiro and C. A. M. Fraga, Am. J. Hypertens., 2010, 23, 135–141 CrossRef CAS PubMed
.
- N. E. Jackson, B. M. Savoie, K. L. Kohlstedt, M. Olvera de la Cruz, G. C. Schatz, L. X. Chen and M. A. Ratner, J. Am. Chem. Soc., 2013, 135, 10475–10483 CrossRef CAS PubMed
.
- C. Chan, A. D. Borthwick, D. Brown, C. L. Burns-Kurtis, M. Campbell, L. Chaudry, C. Chung, M. A. Convery, J. N. Hamblin, L. Johnstone, H. A. Kelly, S. Kleanthous, A. Patikis, C. Patel, A. J. Pateman, S. Senger, G. P. Shah, J. R. Toomey, N. S. Watson, H. E. Weston, C. Whitworth, R. J. Young and P. Zhou, J. Med. Chem., 2007, 50, 1546–1557 CrossRef CAS PubMed
.
- C. A. M. Fraga and E. J. Barreiro, Curr. Med. Chem., 2006, 13, 167–198 CrossRef CAS PubMed
.
- C. Duarte, E. Barreiro and C. Fraga, Mini-Rev. Med. Chem., 2007, 7, 1108–1119 CrossRef CAS PubMed
.
- R. D. C. Maia, R. Tesch and C. A. M. Fraga, Expert Opin. Ther. Pat., 2014, 24, 1161–1170 CrossRef CAS PubMed
.
- F. Weinhold and C. R. Landis, Chem. Educ. Res. Pract., 2001, 2, 91–104 RSC
.
-
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 09, Revision D.01, Gaussian, Inc., Wallingford CT, 2013 Search PubMed
.
- A. D. Becke, Phys. Rev. A: At., Mol., Opt. Phys., 1988, 38, 3098–3100 CrossRef CAS
.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785–789 CrossRef CAS
.
- J. Tomasi, B. Mennucci and R. Cammi, Chem. Rev., 2005, 105, 2999–3094 CrossRef CAS PubMed
.
- B. Mennucci, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 386–404 CrossRef CAS
.
- M. H. Jamróz, Spectrochim. Acta, Part A, 2013, 114, 220–230 CrossRef PubMed
.
- R. Ghosh and S. H. Simonsen, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1993, 49, 1031–1032 CrossRef
.
- H. Gökce and S. Bahçeli, J. Mol. Struct., 2011, 1005, 100–106 CrossRef
.
- J. J. P. Stewart, J. Mol. Model., 2007, 13, 1173–1213 CrossRef CAS PubMed
.
- A. E. Kümmerle, J. M. Raimundo, C. M. Leal, G. S. da Silva, T. L. Balliano, M. A. Pereira, C. A. de Simone, R. T. Sudo, G. Zapata-Sudo and C. A. M. Fraga, Eur. J. Med. Chem., 2009, 44, 4004–4009 CrossRef PubMed
.
- H. Gökce and S. Bahçeli, J. Mol. Struct., 2011, 1005, 100–106 CrossRef
.
- C. M. Breneman and K. B. Wiberg, J. Comput. Chem., 1990, 11, 361–373 CrossRef CAS
.
- P. Politzer, J. S. Murray and T. Clark, Top. Curr. Chem., 2014, 19–42 CrossRef
.
- P. Lorente, I. G. Shenderovich, N. S. Golubev, G. S. Denisov, G. Buntkowsky and H. Limbach, Magn. Reson. Chem., 2001, 39, S18–S29 CrossRef CAS
.
- D. D. Muccio, W. G. Copan, W. W. Abrahamson and G. D. Mateescu, Org. Magn. Reson., 1984, 22, 121–124 CrossRef CAS
.
- M. Delarmelina, G. B. Ferreira, V. F. Ferreira and J. W. D. M. Carneiro, Vib. Spectrosc., 2016, 86, 311–323 CrossRef CAS
.
- Z. Wang, D. Xie, X. Gan, S. Zeng, A. Zhang, L. Yin, B. Song, L. Jin and D. Hu, Bioorg. Med. Chem. Lett., 2017, 27, 4096–4100 CrossRef CAS PubMed
.
- O. Thach, M. Mielczarek, C. Ma, S. K. Kutty, X. Yang, D. S. Black, R. Griffith, P. J. Lewis and N. Kumar, Bioorg. Med. Chem., 2016, 24, 1171–1182 CrossRef CAS PubMed
.
- L. N. F. Cardoso, M. L. F. Bispo, C. R. Kaiser, J. L. Wardell, S. M. S. V. Wardell, M. C. S. Lourenço, F. A. F. M. Bezerra, R. P. P. Soares, M. N. Rocha and M. V. N. de Souza, Arch. Pharm., 2014, 347, 432–448 CrossRef CAS PubMed
.
- T. F. Silva, W. Bispo Júnior, M. S. Alexandre-Moreira, F. N. Costa, C. Monteiro, F. Furlan Ferreira, R. C. R. Barroso, F. Noël, R. T. Sudo, G. Zapata-Sudo, L. M. Lima and E. Barreiro, Molecules, 2015, 20, 3067–3088 CrossRef PubMed
.
- D. J. Van Dijken, P. Kovaříček, S. P. Ihrig and S. Hecht, J. Am. Chem. Soc., 2015, 137, 14982–14991 CrossRef CAS PubMed
.
- C. A. M. Fraga and E. J. Barreiro, Curr. Med. Chem., 2006, 167–198 CrossRef CAS
.
- A. G. Silva, G. Zapata-Sudo, A. E. Kummerle, C. A. M. Fraga, E. J. Barreiro and R. T. Sudo, Bioorg. Med. Chem., 2005, 13, 3431–3437 CrossRef CAS PubMed
.
- G. A. P. Silva, A. E. Kummerle, F. Antunes, C. A. M. Fraga, E. J. Barreiro, G. Zapata-Sudo and R. T. Sudo, Braz. J. Med. Biol. Res., 2013, 46, 263–269 CrossRef CAS PubMed
.
- S. L. Pereira, A. E. Kummerle, C. A. M. Fraga, E. J. Barreiro, R. T. Sudo and G. Zapata-Sudo, Fundam. Clin. Pharmacol., 2014, 28, 29–41 CrossRef CAS PubMed
.
- S. L. Pereira, A. E. Kummerle, C. A. M. Fraga, E. J. Barreiro, N. D. N. Rocha, E. B. Ferraz, J. H. M. Do Nascimento, R. T. Sudo and G. Zapata-Sudo, Eur. J. Pharmacol., 2013, 702, 316–322 CrossRef CAS PubMed
.
- J. A. P. Sato, F. N. Costa, M. D. da Rocha, E. J. Barreiro, C. A. M. Fraga, F. Punzo and F. F. Ferreira, CrystEngComm, 2015, 17, 165–173 RSC
.
- E. J. Barreiro, A. E. Kümmerle and C. A. M. Fraga, Chem. Rev., 2011, 111, 5215–5246 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available: Validation process of the theoretical methodology for the description of the 1,4-N⋯S σ-hole intramolecular interaction. Theoretical and experimental characterization of the 1,4-N⋯S σ-hole intramolecular interaction for the compounds 2-(thiophen-2-yl)pyridine (3), 2-(thiophen-3-yl)pyridine (6), LASSBio-785 (4) and LASSBio-1289 (5). See DOI: 10.1039/c7nj03543h |
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 |