Microfluidic approaches for probing amyloid assembly and behaviour
Abstract
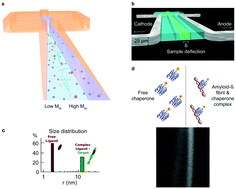
The self-assembly of proteins into supramolecular structures and machinery underpins biological activity in living systems. Misassembled, misfolded and aggregated protein structures can, by contrast, have deleterious activity and such species are at the origin of a number of disease states ranging from cancer to neurodegenerative disorders. In particular, the formation of highly ordered protein aggregates, amyloid fibrils, from normally soluble peptides and proteins, is the common pathological hallmark of a range a group of over fifty protein misfolding disorders. Because of the critical role of the process in the aetiology of such disorders, as well as the quest to understand the basic principles of protein folding and misfolding, the amyloid phenomenon has become a central area of modern biomedical research. Advances in our knowledge of the physical properties of amyloid systems have, however, also highlighted the potential of amyloid structures in the context of materials science. In this review, we explore how microfluidic approaches can be used to study aspects of amyloid assembly and behaviour that are challenging to probe under bulk solution conditions. We discuss the use of volume confinement to probe very early events in the amyloid formation process. In addition, the well-defined fluid flow properties within channels with dimensions on the micron scale can be exploited to measure the physical properties of protein aggregates, such as their sizes and charges, to shed light on the physical and chemical parameters defining amyloid species. Moreover, the molecular species formed during aggregation reactions have physical dimensions spanning at least three orders of magnitude, and microfluidic techniques are well suited to work with analytes of such disparate dimensions. Furthermore, the flexibility of the design of microfluidic devices lends itself to adaptable experimental setups, including the study of protein self-assembly within living cells. Finally, we highlight the salient features of microfluidic experiments that facilitate probing complex biological systems, and discuss their use in the exploration of amyloids as a class of functional material.
- This article is part of the themed collections: Lab on a Chip Recent Review Articles and 7th EuCheMS Chemistry Congress – Molecular frontiers and global challenges


 Please wait while we load your content...
Please wait while we load your content...