Shear dependent red blood cell adhesion in microscale flow†
Abstract
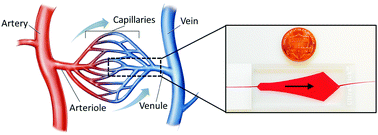
Non-adherence and deformability are the key intrinsic biomechanical features of the red blood cell (RBC), which allow it to tightly squeeze and pass through even the narrowest of microcirculatory networks. Blockage of microcirculatory flow, also known as vaso-occlusion, is a consequence of abnormal cellular adhesion to the vascular endothelium. In sickle cell disease (SCD), an inherited anaemia, even though RBCs have been shown to be heterogeneous in adhesiveness and deformability, this has not been studied in the context of physiologically relevant dynamic shear gradients at the microscale. We developed a microfluidic system that simulates physiologically relevant shear gradients of microcirculatory blood flow at a constant single volumetric flow rate. Using this system, shear dependent adhesion of RBCs from 28 subjects with SCD and from 11 healthy subjects was investigated using vascular endothelial protein functionalized microchannels. We defined a new term, RBC Shear Gradient Microfluidic Adhesion (SiGMA) index to assess shear dependent RBC adhesion in a subject-specific manner. We have shown for the first time that shear dependent adhesion of RBCs is heterogeneous in a microfluidic flow model, which correlates clinically with inflammatory markers and iron overload in subjects with SCD. This study reveals the complex dynamic interactions between RBC-mediated microcirculatory occlusion and clinical outcomes in SCD. These interactions may also be relevant to other microcirculatory disorders and microvascular diseases.


 Please wait while we load your content...
Please wait while we load your content...
