Using an environmentally-relevant panel of Gram-negative bacteria to assess the toxicity of polyallylamine hydrochloride-wrapped gold nanoparticles†
Abstract
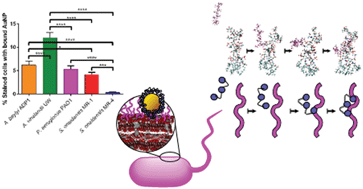
We aim to establish the effect of environmental diversity in evaluating nanotoxicity to bacteria. We assessed the toxicity of 4 nm polyallylamine hydrochloride-wrapped gold nanoparticles (PAH AuNPs) to a panel of bacteria from diverse environmental niches. The bacteria experienced a range of toxicities as evidenced by the different minimum bactericidal concentrations determined; the sensitivities of the bacteria was A. vinelandii = P. aeruginosa > S. oneidensis MR-4 > A. baylyi > S. oneidensis MR-1. Interactions between gold nanoparticles and molecular components of the cell wall were investigated by TEM, flow cytometry, and computational modeling. Binding results showed a general trend that bacteria with smooth lipopolysaccharides (LPS) bind more PAH AuNPs than bacteria with rough LPS. Computational models reveal that PAH migrates to phosphate groups in the core of the LPS structure. Overall, our results demonstrate that simple interactions between nanoparticles and the bacterial cell wall cannot fully account for observed trends in toxicity, which points to the importance of establishing more comprehensive approaches for modeling environmental nanotoxicity.
- This article is part of the themed collections: Best Papers 2018 – Environmental Science: Nano and Celebrating our 2018 prize and award winners


 Please wait while we load your content...
Please wait while we load your content...