Open Access Article
Open Access ArticlePredicting global scale exposure of humans to PCB 153 from historical emissions†
Michael S.
McLachlan
 *a,
Emma
Undeman
*a,
Emma
Undeman
 ab,
Fangyuan
Zhao
ab,
Fangyuan
Zhao
 a and
Matthew
MacLeod
a and
Matthew
MacLeod
 a
a
aDepartment of Environmental Science and Analytical Chemistry (ACES), Stockholm University, 106 91 Stockholm, Sweden. E-mail: michael.mclachlan@aces.su.se
bBaltic Sea Centre, Stockholm University, 106 91 Stockholm, Sweden
First published on 9th March 2018
Abstract
Predicting human exposure to an environmental contaminant based on its emissions is one of the great challenges of environmental chemistry. It has been done successfully on a local or regional scale for some persistent organic pollutants. Here we assess whether it can be done at a global scale, using PCB 153 as a test chemical. The global multimedia fate model BETR Global and the human exposure model ACC-HUMAN were employed to predict the concentration of PCB 153 in human milk for 56 countries around the world from a global historical emissions scenario. The modeled concentrations were compared with measurements in pooled human milk samples from the UNEP/WHO Global Monitoring Plan. The modeled and measured concentrations were highly correlated (r = 0.76, p < 0.0001), and the concentrations were predicted within a factor of 4 for 49 of 78 observations. Modeled concentrations of PCB 153 in human milk were higher than measurements for some European countries, which may reflect weaknesses in the assumptions made for food sourcing and an underestimation of the rate of decrease of concentrations in air during the last decades. Conversely, modeled concentrations were lower than measurements in West African countries, and more work is needed to characterize exposure vectors in this region.
Environmental significanceThis work shows that mathematical models can be used to predict the environmental fate and exposure of a persistent organic pollutant along the whole journey from emissions to human tissues on a global scale with reasonable accuracy. This increases our confidence in our knowledge of the key processes governing the environmental chemistry of such contaminants and our ability to synthesize this knowledge in models. In addition, the work identifies several areas where further work is needed: food sourcing patterns in regions with strong gradients in environmental contamination, exposure pathways to PCBs in Africa, and evaluation of the ability of the models to predict long term time trends. |
Introduction
One of the ultimate goals of environmental chemistry is to be able to predict human exposure to an environmental contaminant based on its emissions. We do this by studying the processes that govern the behaviour of contaminants in different parts of the environment and along different exposure vectors. We then take our knowledge and synthesize it into process-based mathematical models. This is a particularly challenging undertaking for persistent semivolatile chemicals, as they become widely distributed in the environment and also move between and accumulate in different environmental media. To meet this challenge, multimedia fate and transport models have been created that describe how such chemicals become distributed in different parts of our environment following emission.1 We have also developed exposure models that synthesize our understanding of processes that convey chemicals along exposure vectors from environmental media such as air, water and soil to humans.2 In some cases multimedia fate and transport models have been combined with exposure models to create tools that allow us to describe the chemical's complete journey from emission to human tissue.3–7A key activity in this scientific effort is the comparison of the models' estimations against empirical observations. Through such comparisons we can gain confidence that we have synthesized existing knowledge correctly and that the existing knowledge is sufficient to describe key aspects of the transport and environmental fate of persistent semivolatile chemicals and human exposure to them through multiple exposure pathways.
Polychlorinated biphenyls (PCBs) have served as model chemicals in this field of research. Model evaluation for PCBs has been facilitated by the availability of a large amount of measurement data compiled by national, regional and global monitoring programs,8 and the estimates of global PCB emissions to the environment derived from information on PCB production and usage by Knut Breivik and co-workers.9,10 A favourable comparison of PCB concentrations in air measured in Europe and the Arctic versus modeled using the hemispheric fate and transport model MSCE-POP with the “higher scenario” from Breivik's emission estimates provided confidence in both the emissions scenario and the ability of multimedia fate and transport models to explain concentrations in air at a large scale.11 This confidence was further strengthened when good agreement was found between PCB concentrations in air modeled on a global scale with the model BETR Global and measurements.12 The human exposure model ACC-HUMAN has been shown to predict PCB concentrations in food and human milk in Sweden from time trends of measured concentrations in air and water.2 The coupled multimedia fate and exposure model CoZMoMAN was used to predict PCB behaviour in the western Baltic Sea drainage basin and reasonable agreement was found between measured PCB concentrations in most environmental media as well as in food and human milk.7 The same model combination provides good estimates of PCB levels in Norwegians, effectively capturing the influence of different dietary habits and exposure history on concentration in human plasma.13,14 Further confidence was gained when coupled use of the global multimedia fate model GloboPOP and ACC-HUMAN yielded good agreement with measured concentrations of PCBs in human milk in the Arctic for some diet scenarios.15 In summary, research to date has provided evidence that models can predict PCB behaviour in the physical environment at the global scale and human exposure to PCBs in Scandinavia and the Arctic.
In this work we explore whether models can predict human exposure to PCBs on a global scale from historical emissions estimates. This is done using PCB 153, a well-studied congener for which many data are available. We employ a global fate model (BETR Global) together with the ACC-HUMAN human exposure model to predict the concentration of PCB 153 in women from countries around the world. We compare these modeled concentrations with measured concentrations of PCB 153 in pooled human milk samples collected and analysed in the WHO/UNEP global monitoring program.
Methods
The models
The concentrations and fugacities of PCB 153 in the physical environment were modeled with a modified version of the global multimedia fate and transport model BETR Global.16 BETR Global divides the globe into grid cells, and chemical fate in each grid cell is described using a multimedia model. The multimedia model consists of the following compartments: upper atmosphere (representing the free troposphere), lower atmosphere (representing the boundary layer), vegetation, soil, freshwater, freshwater sediment, and ocean water. Exchange between the grid cells occurs via flow of air and water between the upper atmosphere, lower atmosphere, freshwater and ocean water compartments. The original model has a grid cell resolution of 15° × 15°, amounting to 288 cells for the globe. However, because many of the countries that we wished to model were markedly smaller than this, we chose to use a recently created version of this model with a higher spatial resolution. Each cell in the original model was divided into 16 new cells, giving a resolution of 3.75° × 3.75°.17 Some environmental parameters were reassigned with the meteorological models MPI-ESM and BCCR-BCM 2.0. Chemical transport in association with particles from the soil compartment to the freshwater compartment was excluded in the high resolution model as the parameterization was inappropriate for arid environments. The mass transfer coefficients for sediment deposition, sediment resuspension and sediment burial were corrected for a transcription error to 5 × 10−8, 2.5 × 10−8 and 2.5 × 10−8 m h−1, respectively.18 In addition, modifications to the description of atmospheric particle scavenging and oceanic particle organic carbon were made based on recommendations from Trevor Brown as described in the ESI (Text S1†).The exposure of humans to PCB 153 in air, water and soil was calculated using the food chain model ACC-HUMAN.2 ACC-HUMAN uses mechanistically based bioaccumulation models of fish, grass, beef cattle and milk cows. Depending on the organism, the uptake mechanisms considered include ingestion of food, ingestion of soil, inhalation, gill ventilation, drinking, atmospheric deposition and root uptake while the elimination pathways treated are egestion, urination, exhalation, gill ventilation, lactation and metabolism. Temporal variation in physiological and environmental parameters as well as growth are taken into account. The bioaccumulation models predict chemical residues in fish, beef and milk arising from contamination in water, air and soil. The concentrations in these foodstuffs, water and air are then input into a mass balance model of humans which considers elimination via metabolism, exfoliation, digestive tract excretion, exhalation, childbirth and nursing and calculates the chemical concentrations in human tissue and milk. The human sub-model is non-steady state and considers changes in diet and body lipid mass with age as well as chemical elimination via birth and nursing.
BETR Global was run for the period 1930 until 2015. For each country considered in this study, the fugacities of PCB 153 in the lower atmosphere, freshwater and soil compartments during the simulation period were selected from the model output as well as the temperatures in the air and freshwater compartments and the organic carbon content of the soil. In those cases where the country was located in more than one grid cell, a population-weighted average of these parameters for all of the country's grid cells was calculated. For this purpose population density data with a resolution of 30 × 30 arc seconds from the Center for International Earth Science Information Network were regridded onto the 3.75° × 3.75° resolution of the BETR Global model.19 The fugacities, temperatures and soil organic carbon content were input into ACC-HUMAN and the concentrations in humans arising from all sources of exposure except marine fish were calculated. The ACC-HUMAN output selected was the concentration in human milk three months after childbirth, in a woman of the average age of primiparae mothers in the country being modeled, in the year when the human milk samples for that country were collected. A separate ACC-HUMAN simulation was done for exposure to PCB 153 from consumption of marine fish only (a description of the grid averaging of the BETR Global output for this simulation is given below) and the result was added to the first simulation to obtain the concentration of PCB 153 in human milk from all sources of exposure. Some sensitivity modeling was performed, the details of which are provided upon mention below.
Parameterization
The historical global PCB emission scenarios produced by Breivik et al. formed the basis for the simulations. The scenarios consider emissions during production and during the full life cycle of products containing PCBs.10 They were recently updated to include emissions arising from e-waste export/import.20 We primarily used the data for the “default scenario” but also did a comparison with the “worst-case scenario”.21 Both of these scenarios build on the previous “higher scenario”. They differ from each other in the fraction of imported e-waste that is assumed to be burned (5% vs. 20%).20 In addition to new emissions from e-waste trade, the updated scenarios include emissions from production and formulation that were not included in the earlier scenarios.22 These new emissions occur primarily during the 1950s and 1960s, and their inclusion increased the total global historical emissions of PCB 153 from 2600 tonnes to 4700 tonnes. The emissions scenario data, which are provided on a 1° × 1° resolution, were regridded to match the 3.75° × 3.75° resolution of the revised BETR Global model. All emissions were into the lower atmosphere compartment of the respective grid cell. Secondary emissions are not included in the emissions scenario as they are generated in the BETR-Global model.The physical chemical properties of PCB 153 are listed in Table S1.† The partitioning properties were taken from Schenker et al.23 The degradation half-lives in water, soil, and sediment were taken from Wania and Daly, while the degradation half-life in air was taken from Anderson and Hites.24,25
For bioaccumulation in the aquatic and agricultural food chains, the parameterization in ACC-HUMAN as defined for the temperate region in Undeman et al. was used for all countries.26 However, country-specific values were used for temperatures and human dietary intake. Temperature and human diet composition were shown to be the ecosystem properties having the largest influence on human exposure in a comparative study of humans living in different climate zones and consuming only regionally produced food.26 The dietary intake of different food items was taken from the WHO Global Environment Modeling System (GEMS) cluster diets. GEMS has defined 17 cluster diets for the globe and specified for each country which cluster diet best represents food consumption in that country.27 The wet weight consumption rates of each food item in the cluster diets was converted to a lipid consumption rate using lipid contents derived from the FAO's food balance sheets.28 All animal-based food items were then grouped into the ACC-HUMAN categories meat, dairy products (including eggs), freshwater fish and marine fish, and the total lipid consumption rate for each category was determined (see Text S2 and Tables S2–S4† for more details). These were used to define the dietary intake of a 25 year old woman in ACC-HUMAN, which was assumed to be constant over time. The dietary intake at 25 years of age was converted into a lifetime dietary intake curve using the default functions in ACC-HUMAN. The dietary intake of animal lipid by a 25 year old woman ranged from 5 g d−1 (some countries in central Africa) to 85 g d−1 (some countries in central Europe).
Food sourcing was also country specific. It was assumed that meat, dairy products and freshwater fish were produced within the country, but that marine fish were harvested internationally. The marine fish sourcing was the same for all countries. It was based on a map of global fish harvesting.29 The fugacity in water for the marine fish simulation was the average of the fugacity in all BETR Global grid cells that had the highest fish harvesting rate (0.18–520 ton km−2) on this map. Since both the dietary intake of marine fish and the age of the primiparae mothers was country specific, a separate ACC-HUMAN simulation of human exposure via marine fish was done for each country.
Age is known to influence the concentration of persistent contaminants in humans during post-ban periods when exposure is decreasing.30 Therefore country specific data on the typical age of primiparae women were used.31
Human milk data
The model simulations were compared with PCB 153 concentrations measured in human milk under the WHO/UNEP global monitoring plan for persistent organic pollutants under the Stockholm Convention. This data source was chosen because of the excellent quality assurance and the internal consistency of the data.32 PCB 153 was chosen because it is the PCB congener for which the most data were available. Data were taken for samples collected between 2000 and 2014, comprising the 3rd and later WHO surveys. Between 2000 and 2003 pools of milk from 10 donors were analysed; thereafter the milk from at least 50 donors was pooled and the donors were considered to represent the background contamination situation in the country.32 Inclusion criteria for donating mothers were that they were primiparae, healthy, nursing one child, and had been a local resident for at least 5 years. The data were downloaded from a data repository operated by Masaryk University.33 A total of 94 data points from 69 countries were available. Of these, 13 countries (16 data points) were excluded because the countries were too small to be resolved by the spatial scale of the global model (Antigua and Barbuda, Barbados, Fiji, Jamaica, Kiribati, Marshall Islands, Mauritius, Niue, Palau, Samoa, Solomon Islands, Tonga and Tuvalu); BETR Global does not include land compartments in a model cell when the land surface area is very small. The remaining countries were distributed across all continents with the strongest representation in Europe (22) and Africa (14). A total of 24 data points from the 1st and 2nd WHO surveys in 1987 and 1992 were not included because of higher sampling and analytical uncertainty and a strong regional bias (19 of 24 data points from European countries).Statistics
Correlation and regression analysis was carried out using JMP Pro 13.1.0.Results and discussion
We first evaluate the ability of the model to predict changes in the concentrations in human milk over time before comparing measured and modeled concentrations in space and assessing the model accuracy.Variability of PCB 153 concentrations in human milk over time
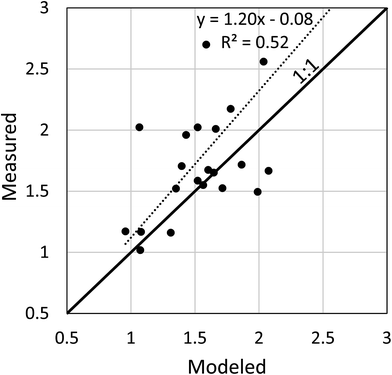
Measured data were available for more than one year for 20 countries. In all cases but one a decrease in the concentration of PCB 153 in human milk was observed (Table S5†). To evaluate the ability of the model to capture the temporal trend in the measured data, the quotient of the concentrations between the two sampling years was calculated. These quotients obtained for the measured data and modeled data are compared in Fig. 1. The measured and the modeled quotients were highly correlated (r = 0.72, p = 0.0005). However, the modeled decrease in concentration tended to be slower than the observed decrease; for 15 of 22 data points (14 of 20 countries) the modeled decrease was slower with a median residual of 0.33, while for 7 data points the modeled decrease was faster with a median residual of 0.17. This tendency to under-predict the decrease in concentration in human milk could be due to an underestimation of the rate of decrease of emissions, an underestimation of the rate of removal of PCBs in the environment, or changes over time in the exposure vectors such as dietary habits or food sourcing that were not considered in the model.The under-prediction of the decrease in concentrations in human milk must be due to either weaknesses in the emissions/fate modeling or in the bioaccumulation/exposure modeling. To assess the former, we compared modeled time trends for concentrations in air with time trends constructed from measured PCB 153 concentrations in air reported in the data repository for the WHO/UNEP global monitoring plan.33 We note that the concentration in air controls exposure via the terrestrial food chain and indirectly influences exposure via the aquatic food web. We extracted passive air sampling data that had been collected at the same location in 3 or more different years. The earliest data used were from 2005, the most recent from 2014. Details of the data selection are provide in Text S3 in the ESI.† A total of 58 data sets were extracted and linear regressions of ln concentration vs. year were made to determine the first order time constant. The regressions yielded an r2 > 0.5 for 40 of the data sets; 36 of these were for sites in Europe. The time constants for these 40 data sets were compared with time constants calculated from modeled values of the average annual concentration of PCB 153 in air for the same location and time period. In all but two cases the measured time constant was more negative, i.e., greater than the modeled time constant (see Fig. 2). The measured time constant was on average 4.4 (median 3.6) times greater than the modeled. This suggests that under-prediction of the rate of decrease of environmental concentration by the emissions/fate modeling was one of the reasons for the under-prediction of the rate of decrease in the concentration of PCB 153 in human milk.
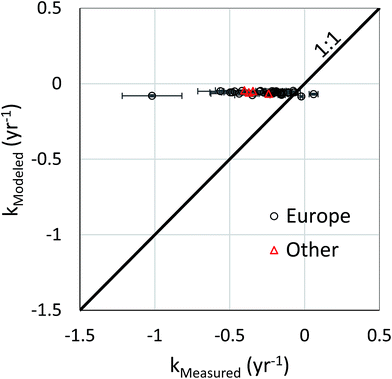 | ||
| Fig. 2 Modeled versus measured time constant for the change in the concentration of PCB 153 in air. The measured data were derived from passive samplers deployed at 40 sites around the world (36 of them in Europe) for varying time periods between 2005 and 2014. The modeled time constants were derived from modeled average annual concentrations in air for the same site and for the same time period as the measured data. The mean and standard error of the time constant are shown. The thick line shows the line of perfect agreement between the modeled and measured values. For an expanded image of the data, see Fig. S2.† | ||
Several authors have reported a tendency of model scenarios to underestimate the rate of decline of concentrations of PCB 153 in air. Hauck et al. compared modeled and measured time constants for 3 sites in Europe for the period 1994–2002 and obtained similar results to ours: modeled values of −0.06, −0.06 and −0.07 per year compared to measured values of −0.48, −0.17 and −0.17 per year.34 Breivik et al. observed that the modeled rate of decline was slower than the measured rate of decline for sites in Sweden and Norway during the period 1989–2005 and proposed deficits in the historical emissions scenario as a possible weakness in their chain of models.7 In Breivik and co-workers' most recently published PCB emissions scenario an effort was made to address these deficits; emissions prior to 1984 were increased by 123%, resulting in a considerably steeper decline in PCB emissions between 1976 and 1984.20,21 We used this emissions scenario in our model.
On the other hand, there are studies showing rates of decline of concentrations in air that are more consistent with the model predictions (0.05–0.08 per year, Fig. 2 and S2†). Active air sampling at a semi-rural site in England between 1992 and 2007 yielded a time constant of 0.14 per year,35 while passive sampling at rural sites in Wales and Scotland between 1998 and 2008 yielded time constants of 0.08 and 0.12 per year, respectively.36 Several active sampling studies providing only data for the sum of the indicator PCBs have also reported low rates of decline: 0.09 per year for a semi-rural site in England (1992–2012), 0.12 per year for several rural sites in England (1997–2012),37 and 0.09 per year for 8 EMEP sites located in Scandinavia (mid 1990's–2008).35 The variability in the reported empirical observations makes it difficult to establish whether the modeled rate of decline of the concentration of PCB 153 in air is indeed underestimated.
Another potential confounding factor is the change in sampling strategy for the human milk samples. Up until 2003 pools of milk from 10 donors were analysed, while thereafter the milk from at least 50 donors who had been selected to be representative for the country was pooled. Up until 2003 it is not clear if the donors were selected to be representative for the country as a whole, and this together with the smaller number of individuals in the pool makes the representativeness of the pooled sample less certain. There was no spatial trend in the deviation between modeled and measured; in Europe the model over-predicted for Romania, Ireland, Norway and Sweden while it strongly under-predicted (residual > 0.5) for Belgium, Bulgaria, Hungary, The Netherlands and Spain. The dataset thus provides little material for further exploration of the model's tendency to under-predict the decrease in concentration.
Spatial variability of PCB 153 concentrations in human milk and model accuracy
The prediction of spatial variability and model accuracy were assessed by comparing modeled and measured concentrations in human milk using a hierarchy of statistics with increasing explanatory power: range, rank correlation analysis, the Pearson correlation coefficient, and the root mean square difference.The mean concentrations measured in human milk ranged from 0.76 (Ethiopia) to 260 (Czech Republic) ng g−1 lipid. The modeled concentrations ranged from 0.23 (Uganda) to 520 (The Netherlands) ng g−1 lipid. The range of model predictions encompassed the range of measured values.
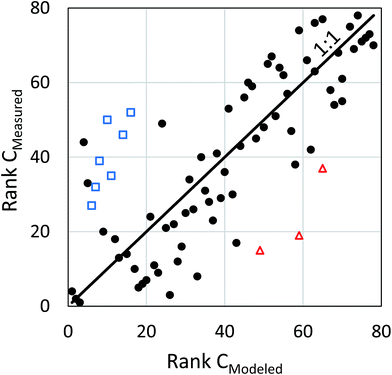
The ability of the models to predict the rank order of the PCB 153 concentrations was tested using the Spearman rank correlation test. There was a strong positive monotonic relationship between the modeled and measured concentration of PCB 153 in human milk (rs = 0.74, n = 78, p < 0.0001). A higher rank for the modeled concentration in the dataset tended to be accompanied by a higher rank for the measured concentration (Fig. 3).
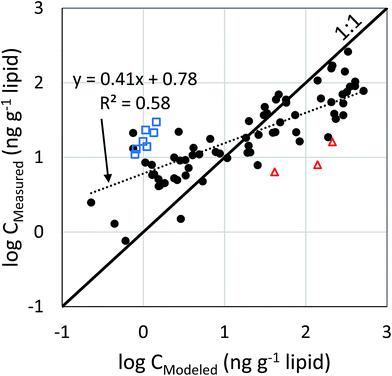
The ability of the model to quantitatively capture the variability in the concentration of PCB 153 in human milk was tested using the Pearson correlation test. The data were log transformed to reduce skewedness. There was a strong correlation between the log modeled concentration and log measured concentration (r = 0.76, n = 78, p < 0.0001). Fig. 4 shows the linear regression of the modeled against the measured concentration. The model was able to explain 58% of the measured variability in the human milk concentration.
The accuracy of the model predictions was assessed using the difference between the logarithms of the measured and modeled concentrations. The root mean square difference was 0.65 log units, which corresponds to a factor of 4.5.
Exploration of the deviations between predictions and observations
The data were studied more closely to identify specific areas of model strength and weakness. To provide orientation for this work, simple regressions were tested to identify the major factors driving the model outcome. A linear regression of the logarithm of the modeled PCB 153 concentration in human milk against the logarithm of the dietary ingestion of lipids of animal origin (which ranged from 5 to 85 g d−1, see above) gave a strong fit (r2 = 0.73, n = 78, p < 0.0001, rms error = 0.49). Selecting the logarithm of the average fugacity of PCB 153 in air in the country 10 years prior to the sampling year as the dependent variable gave a somewhat weaker fit (r2 = 0.42, n = 78, p < 0.0001, rms error = 0.73). Note that the 10 year span between air concentration and human milk sampling was chosen because the PCB 153 concentration in humans is the result of historical exposure. A regression using both dietary ingestion of lipids and fugacity in air as independent variables improved the strength of the fit (r2 = 0.90, n = 78, p < 0.0001, rms error = 0.31). Clearly the modeled fugacity in air and the estimated dietary intake of animal lipids are two major factors determining the model outcome.Comparison of the modeled and measured concentration of PCB 153 in human milk with the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
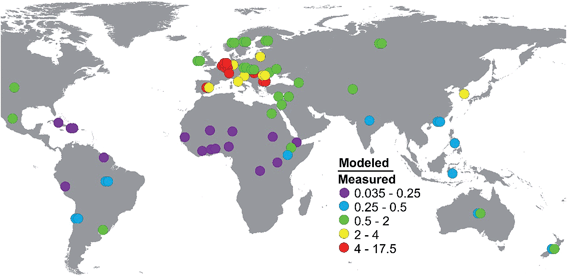
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 line of perfect agreement shows two regions with a bias (Fig. 4). The model under-predicts when the concentration in milk is low, and it over-predicts when the concentrations in milk are high. There is a strong geographic correlation that underlies these biases. Those countries for which the model over-predicts by a factor 4 or more are all located in Europe, whereas those countries for which the model under-predicts by a factor 4 or more are located primarily in Africa (Fig. 5, Table S5†).
1 line of perfect agreement shows two regions with a bias (Fig. 4). The model under-predicts when the concentration in milk is low, and it over-predicts when the concentrations in milk are high. There is a strong geographic correlation that underlies these biases. Those countries for which the model over-predicts by a factor 4 or more are all located in Europe, whereas those countries for which the model under-predicts by a factor 4 or more are located primarily in Africa (Fig. 5, Table S5†).
Among European countries there was a large variability in the model fit. While the model over-predicted by at least a factor of 4 for 7 countries (13 data points), it predicted within a factor of 2 for 11 countries (19 data points, see Fig. 5 and Table S5†). The 7 countries that were strongly over-predicted were the 3 Benelux countries, Switzerland, Spain, Bulgaria and Hungary. Of these, the measured concentrations in the Benelux countries were in the upper range of the measured concentrations in other European countries sampled at the same time. The over-prediction for these countries and good prediction for others suggests that the model predicts stronger gradients between European countries than indicated by the observations.
One possible explanation for the stronger gradients in the modeled concentrations could be the assumption that all of the diet with the exception of marine fish is sourced within the country.38,39 Within the European Union there is intensive trade in agricultural goods. This trade likely reduces the differences in dietary exposure between countries. To test this hypothesis we modeled European milk, meat and freshwater fish using average fugacities for EU countries that were weighted according to the countries' fraction of the permanent pastures and meadows in the EU (see Table S6†). We then assumed that all of dairy, meat and freshwater fish consumed in each EU country were this EU food and recalculated the PCB 153 concentrations in human milk. The results showed an improved model match for the Benelux countries (modeled/measured quotient (Q) changed from 10.2, 7.2, 6.6, 5.9, 5.5, 4.5 and 4.3 to 6.3, 4.5, 4.1, 3.7, 3.5, 2.8 and 2.7). However, there was little or no impact on Hungary, and the fit for Bulgaria worsened (Q from 6.5 and 5.2 to 10.5 and 8.7). In addition, the model now strongly overestimated the concentrations for several countries that had originally had a good fit like Finland (Q from 1.01 and 0.83 to 6.0 and 4.9) and Ireland (Q from 1.94, 1.84 and 1.38 to 13.0, 12.3 and 9.4) (Table S7†). The countries for which the fit worsened were countries that had lower local levels of PCB 153 contamination than the EU average. They tended to lie on the northern and western fringes of Europe, but also included Bulgaria. Central Europe has been a major global source of PCBs,10 and this explains the strong gradients in contamination. In regions with strong gradients in contamination, detailed information on food sourcing is important. Such detailed information on food sourcing in Europe was not available for this study. It is possible that such information would improve the capabilities of models to predict PCB 153 concentrations in human milk.
In addition to the stronger gradients in modeled concentrations among European countries, the modeled concentration for Europe as a whole were higher than measurements. This could be related to the model's tendency to under-predict the decrease in PCB 153 concentrations over time (see above). Since Europe is a former strong source region where PCB concentrations have been declining for the last 4 decades, the modeled concentration in human milk will be particularly sensitive to the model correctly predicting the decrease in PCB concentrations in the environment over time. The amount of lipids per capita in the European food supply has decreased by 10% between 1985 and 2010,28 but this change is small compared to the model over-prediction and thus unlikely to be a major contributing factor.
In 2001 Hungary had the lowest measured PCB 153 concentration in human milk of any of the 19 European countries sampled during 2001–2002. The measured concentration (16 ng g−1 lipid) was 3.4–10 times lower than in neighbouring countries to the north (Slovakia, 170 ng g−1 lipid), east (Ukraine, 54 ng g−1 lipid; Romania, 62 ng g−1 lipid) and south (Croatia, 54 ng g−1 lipid). We can offer no hypothesis as to why concentrations in Hungary should be so much lower than in surrounding countries (emissions were similar). The measured concentration in Bulgaria in 2001 was only marginally higher than in Hungary.
Turning to the systematic under-prediction of low PCB 153 concentrations, the 10 countries showing the smallest ratio of modeled![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) measured concentration were from Africa, 7 of them from West Africa (Fig. 4, Table S5†). In addition, 4 of the 7 samples showing the largest difference in rank order (from 31 to 40 places) were from West Africa (all ranked too low), with the others coming from Hungary, Bulgaria and Sudan (Fig. 3). A possible explanation for the model's under-estimation in the West African countries could be an under-estimation of PCB emissions. Measurements of PCB concentrations in West African air show elevated levels compared to the rest of the world. This has been attributed to emissions arising from e-waste management and other waste related issues.40–44 Breivik and co-workers concluded that PCB emissions had been underestimated in regions that have received e-waste and other waste products containing PCBs such as West Africa, and revised their emissions estimates.20,42 In our simulations we have used their revised “default” emission scenario, which is based on the assumption that 5% of the disposed e-waste in e-waste importing countries is subject to open burning.20 Although this scenario takes into consideration emissions from e-waste management in West Africa, the PCB 153 concentrations in human milk were still strongly under-predicted. We therefore re-did the model simulations using the “worst case” scenario assembled by Breivik and co-workers, which assumes that 20% of the disposed e-waste is subject to open burning.20 The impact on the ratio of modeled
measured concentration were from Africa, 7 of them from West Africa (Fig. 4, Table S5†). In addition, 4 of the 7 samples showing the largest difference in rank order (from 31 to 40 places) were from West Africa (all ranked too low), with the others coming from Hungary, Bulgaria and Sudan (Fig. 3). A possible explanation for the model's under-estimation in the West African countries could be an under-estimation of PCB emissions. Measurements of PCB concentrations in West African air show elevated levels compared to the rest of the world. This has been attributed to emissions arising from e-waste management and other waste related issues.40–44 Breivik and co-workers concluded that PCB emissions had been underestimated in regions that have received e-waste and other waste products containing PCBs such as West Africa, and revised their emissions estimates.20,42 In our simulations we have used their revised “default” emission scenario, which is based on the assumption that 5% of the disposed e-waste in e-waste importing countries is subject to open burning.20 Although this scenario takes into consideration emissions from e-waste management in West Africa, the PCB 153 concentrations in human milk were still strongly under-predicted. We therefore re-did the model simulations using the “worst case” scenario assembled by Breivik and co-workers, which assumes that 20% of the disposed e-waste is subject to open burning.20 The impact on the ratio of modeled![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) measured concentrations was small (an increase from 0.046–0.079 to 0.052–0.102 for the 7 West African countries, see Table S8†). This indicates that emissions to air resulting from e-waste management followed by regional scale uptake in milk, meat and freshwater fish could not explain the model's under-prediction in West Africa. Alternative explanations could be contamination of these foodstuffs occurring at a more local scale not captured by the model (the spatial resolution of the BETR Global model was 3.75° × 3.75°) or other exposure vectors related to emissions from e-waste management.
measured concentrations was small (an increase from 0.046–0.079 to 0.052–0.102 for the 7 West African countries, see Table S8†). This indicates that emissions to air resulting from e-waste management followed by regional scale uptake in milk, meat and freshwater fish could not explain the model's under-prediction in West Africa. Alternative explanations could be contamination of these foodstuffs occurring at a more local scale not captured by the model (the spatial resolution of the BETR Global model was 3.75° × 3.75°) or other exposure vectors related to emissions from e-waste management.
Another contributing factor to the difference between the modeled and observed concentrations could be the representativeness of the pooled human milk samples that were analyzed. Socioeconomic factors such as gross national income per capita and urban versus rural residence have recently been found to influence PCB levels in people from West Africa.45 Differences in diet are a possible link between these socioeconomic factors and PCB levels. Furthermore, PCB levels in people from West African countries that are major importers of e-waste are higher than in people from other West African countries.42 This suggests that there could be strong gradients in exposure within a country, and this would have to be taken into consideration in assembling representative pooled samples.
Three other African countries, Democratic Republic of Congo, Uganda and Sudan, were also strongly under-predicted by the model (Fig. 5, Table S5†). Elevated PCB emissions from e-waste management in West Africa are unlikely to explain the discrepancy in these countries. A common feature of both the East African and West African countries was low dietary ingestion of lipids of animal origin, from 5.8 to 10.5 g d−1, as reported in the GEMS cluster diets. This compares with 56–84 g d−1 for most European countries. As discussed above, the dietary ingestion of lipids of animal origin is a strong predictor of the model outcome. It is conceivable that other sources of exposure to PCB 153 are important in countries where the exposure via lipids of animal origin is comparatively low. However, the agreement between the modeled and measured concentrations was good to very good for two other East African countries, Kenya and Ethiopia (P/O of 0.34 and 0.79), so no systematic trend was present to support this hypothesis.
Another common feature of the African countries was that marine fish accounted for >50% of PCB exposure. For our simulations we have assumed that marine fish are sourced from an international pool. In reality, a significant portion of the fish consumed in these countries is likely to be sourced from local or regional coastal waters. As a first assessment of the possible impact of this assumption, we assumed that the fugacity in the coastal waters was equal to the fugacity in freshwater for a given country, and compared this with the fugacity in marine water for the global marine fish simulation. For the African countries the approximated fugacity in coastal water was similar to or lower than the fugacity in marine water. Hence this assessment provided no indication that the assumption of global sourcing of marine fish could explain the strong underestimation of concentrations in human milk in Africa. However, this assessment does not consider other possible vectors such as emissions of PCBs to coastal waters as a result of e-waste management practices.
The relatively poor agreement between the models and observations for many African countries highlights the need for a better understanding of environment levels and exposure vectors for PCBs in this region. There is comparatively little information available, despite indications that high exposure is occurring, particularly for sub-groups exposed via emissions related to waste management.
Conclusions
Despite the systematic differences between model predictions and observations discussed above, the concentrations of 49 out of 78 data points were predicted within a factor of 4. This is an impressive result, considering that the model encompasses emissions estimates, environmental fate, bioaccumulation and human exposure on a global scale. The model used here captures major determinants of PCB 153 concentrations in human milk. Further improvements could be achieved by integrating more detailed knowledge of food sourcing in areas with strong spatial gradients in contamination levels. There is also a need to more closely evaluate the ability of the models to predict long-term time trends. Finally, understanding of exposure vectors to chemicals in developing economies is an important knowledge gap. We conclude that integrated modeling of chemical fate, bioaccumulation and human exposure is a useful tool for studying persistent organic pollutants on a global scale. At the same time, we caution that our results do not provide a justification for non-critical application of these modeling tools to different problems.46 Confidence in the predictive ability of a model must be built upon evaluation of those key components of the model that determine the model outcome of interest. In modeling human exposure to chemical contaminants, the key model components can vary widely depending on the scenario and the chemical.Conflicts of interest
The authors have no conflicts of interest.Acknowledgements
We thank Trevor Brown for sharing his modifications to the BETR Global model and the text describing those modifications. We thank Erik Smedberg for contributions to maps produced in Arc GIS. We thank Frank Wania for reading and commenting on an earlier version of the manuscript. FZ and MMa received funding from the Research Council of Norway through the Økosystem project #244298 and from the European Union's Horizon 2020 research and innovation programme under grant agreement No. 689443 via project iCUPE (Integrative and Comprehensive Understanding on Polar Environments). EU acknowledges financial support from the Baltic Sea Adaptive Management (BEAM) project at Stockholm University.References
- M. MacLeod, M. Scheringer, T. E. McKone and K. Hungerbuhler, The state of multimedia mass-balance modeling in environmental science and decision-making, Environ. Sci. Technol., 2010, 44, 8360–8364 CrossRef CAS PubMed.
- G. Czub and M. S. McLachlan, A food chain model to predict the levels of lipophilic organic contaminants in humans, Environ. Toxicol. Chem., 2004, 23, 2356–2366 CrossRef CAS PubMed.
- T. G. Vermeire, D. T. Jager, B. Bussian, J. Devillers, K. Den Haan, B. Hansen, I. Lundberg, H. Niessen, S. Robertson, H. Tyle and P. T. J. van der Zandt, European Union System for the Evaluation of Substances (EUSES). Principles and structure, Chemosphere, 1997, 34, 1823–1836 CrossRef CAS PubMed.
- T. E. McKone and D. W. Layton, Screening the potential risks of toxic-substances using a multimedia compartment model – Estimation of human exposure, Regul. Toxicol. Pharmacol., 1986, 6, 359–380 CrossRef CAS PubMed.
- D. W. Pennington, M. Margni, C. Ammann and O. Jolliet, Multimedia fate and human intake modeling: Spatial versus nonspatial insights for chemical emissions in Western Europe, Environ. Sci. Technol., 2005, 39, 1119–1128 CrossRef CAS PubMed.
- J. A. Arnot, D. Mackay, E. Webster and J. M. Southwood, Screening level risk assessment model for chemical fate and effects in the environment, Environ. Sci. Technol., 2006, 40, 2316–2323 CrossRef CAS PubMed.
- K. Breivik, G. Czub, M. S. McLachlan and F. Wania, Towards an understanding of the link between environmental emissions and human body burdens of PCBs using CoZMoMAN, Environ. Int., 2010, 36, 85–91 CrossRef CAS PubMed.
- United Nations Environment, Global Monitoring Plan for Persistent Organic Pollutants Second Global Monitoring Report under the Stockholm Convention Article 16 on Effectiveness Evaluation, available from: http://chm.pops.int/Implementation/GlobalMonitoringPlan/MonitoringReports/tabid/525/Default.aspx, 2017.
- K. Breivik, A. Sweetman, J. M. Pacyna and K. C. Jones, Towards a global historical emission inventory for selected PCB congeners—a mass balance approach. 2. Emissions, Sci. Total Environ., 2002, 290, 199–224 CrossRef CAS PubMed.
- K. Breivik, A. Sweetman, J. M. Pacyna and K. C. Jones, Towards a global historical mission inventory for selected PCB congeners—A mass balance approach 3. An update, Sci. Total Environ., 2007, 377, 296–307 CrossRef CAS PubMed.
- A. Malanichev, E. Mantseva, V. Shatalov, B. Strukov and N. Vulzkh, Numerical evaluation of the PCBs transport over the Northern Hemisphere, Environ. Pollut., 2004, 128, 279–289 CrossRef CAS PubMed.
- L. Lamon, H. von Waldow, M. MacLeod, M. Scheringer, A. Marcomini and K. Hungebühler, Modeling the global levels and distribution of polychlorinated biphenyls in air under a climate change scenario, Environ. Sci. Technol., 2009, 43, 5818–5824 CrossRef CAS PubMed.
- T. Haugdahl Nøst, K. Breivik, O.-M. Fuskevåg, J. Øyvind Odland and T. Manning Sandanger, Persistent organic pollutants in Norwegian men from 1979 to 2007: Intraindividual changes, age–period–cohort effects, and model predictions, Environ. Health Perspect., 2013, 121, 1292–1298 Search PubMed.
- T. Haugdahl Nøst, K. Breivik, F. Wania, C. Rylander, J. Øyvind Odland and T. Manning Sandanger, Estimating time-varying PCB exposures using person-specific predictions to supplement measured values: A comparison of observed and predicted values in two cohorts of Norwegian women, Environ. Health Perspect., 2016, 124, 299–305 Search PubMed.
- M. J. Binnington, M. S. Curren, H. M. Chan and F. Wania, Balancing the benefits and costs of traditional food substitution by indigenous Arctic women of childbearing age: Impacts on persistent organic pollutant, mercury, and nutrient intakes, Environ. Int., 2016, 94, 554–566 CrossRef CAS PubMed.
- M. MacLeod, H. von Waldow, P. Tay, J. M. Armitage, H. Wöhrnschimmel, W. J. Riley, T. E. McKone and K. Hungerbühler, BETR global – A geographically-explicit global-scale multimedia contaminant fate model, Environ. Pollut., 2011, 159, 1442–1445 CrossRef CAS PubMed.
- R. K. Göktas and M. MacLeod, Remoteness from sources of persistent organic pollutants in the multi-media global environment, Environ. Pollut., 2016, 217, 33–41 CrossRef PubMed.
- D. Mackay, Multimedia environmental models: the fugacity approach, Lewis, Boca Raton, 2nd edn, 2001, p. 178 Search PubMed.
- Center for International Earth Science Information Network – CIESIN – Columbia University, 2016, Gridded Population of the World, Version 4 (GPWv4): Population Density Adjusted to Match 2015 Revision UN WPP Country Totals. Palisades, NY: NASA Socioeconomic Data and Applications Center (SEDAC), DOI:10.7927/H4HX19NJ, accessed October 2017.
- K. Breivik, J. M. Armitage, F. Wania, A. J. Sweetman and K. C. Jones, Tracking the global distribution of persistent organic pollutants accounting for e-waste exports to developing regions, Environ. Sci. Technol., 2016, 50, 798–805 CrossRef CAS PubMed.
- K. Breivik, Emissions data, https://www.nilu.no/projects/globalpcb/, accessed November 2017.
- K. Breivik, Personal communication.
- U. Schenker, M. MacLeod, M. Scheringer and K. Hungerbühler, Improving data quality for environmental fate models: A least squares adjustment procedure for harmonizing physicochemical properties of organic compounds, Environ. Sci. Technol., 2005, 39, 8434–8441 CrossRef CAS PubMed.
- F. Wania and G. L. Daly, Estimating the contribution of degradation in air and deposition to the deep sea to the global loss of PCBs, Atmos. Environ., 2002, 36, 5581–5593 CrossRef CAS.
- P. N. Anderson and R. A. Hites, OH radical reactions:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) The major removal pathway for polychlorinated biphenyls from the atmosphere, Environ. Sci. Technol., 1996, 30, 1756–1763 CrossRef CAS.
The major removal pathway for polychlorinated biphenyls from the atmosphere, Environ. Sci. Technol., 1996, 30, 1756–1763 CrossRef CAS. - E. Undeman, T. N. Brown, F. Wania and M. S. McLachlan, Susceptibility of human populations to environmental exposure to organic contaminants, Environ. Sci. Technol., 2010, 44, 6249–6255 CrossRef CAS PubMed.
- GEMS cluster diets, https://undatacatalog.org/dataset/gemsfood-consumption-database, accessed in September 2017.
- FAO food balance sheets, http://www.fao.org/faostat/en/#data/FBS, accessed in September 2017.
- Map of Global Fishing 2014, The Sea Around Us Research Initiative at The University of British Columbia, http://www.seaaroundus.org/data/#/spatial-catch, accessed in September 2017.
- R. Ritter, M. Scheringer, M. MacLeod, U. Schenker and K. Hungerbühler, A multi-individual pharmacokinetic model framework for interpreting time trends of persistent chemicals in human populations: Application to a postban situation, Environ. Health Perspect., 2009, 117, 1280–1286 CAS.
- The World Factbook, https://www.cia.gov/library/publications/the-world-factbook/fields/2256.html#af, accessed in September 2017.
- M. van den Berg, K. Kypke, A. Kotz, A. Tritscher, S. Y. Lee, K. Magulova, H. Fiedler and R. Malisch, WHO/UNEP global surveys of PCDDs, PCDFs, PCBs and DDTs in human milk and benefit – risk evaluation of breastfeeding, Arch. Toxicol., 2017, 91, 83–96 CrossRef CAS PubMed.
- R. Hulek, J. Boruvkova, J. Gregor, J. Kalina, Z. Bednarova, K. Sebkova, O. Melkes, R. Novak, J. Jarkovsky, L. Dusek and J. Klanova, Global Monitoring Plan of the Stockholm Convention on Persistent Organic Pollutants: visualisation and on-line analysis of global levels of chemicals in air, water, breast milk and blood, http://www.pops-gmp.org/visualization-2014, accessed in September 2014.
- M. Hauck, M. A. J. Huijbregts, A. Hollander, A. J. Hendriks and D. van de Meent, Modeled and monitored variation in space and time of PCB-153 concentrations in air, sediment, soil and aquatic biota on a European scale, Sci. Total Environ., 2010, 408, 3831–3839 CrossRef CAS PubMed.
- J. K. Schuster, R. Gioia, A. J. Sweetman and K. C. Jones, Temporal trends and controlling factors for polychlorinated biphenyls in the UK atmosphere (1991–2008), Environ. Sci. Technol., 2010, 44, 8068–8074 CrossRef CAS PubMed.
- J. K. Schuster, R. Gioia, k. Breivik, E. Steinnes, M. Scheringer and K. C. Jones, Trends in European background air reflect reductions in primary emissions of PCBs and PBDEs, Environ. Sci. Technol., 2010, 44, 6760–6766 CrossRef CAS PubMed.
- C. Graf, A. Katsoyiannis, K. C. Jones and A. J. Sweetman, The TOMPs ambient air monitoring network – Continuous data on UK air quality for over 20 years, Environ. Pollut., 2016, 217, 42–51 CrossRef CAS PubMed.
- M. MacLeod, D. H. Bennett, M. Perem, R. L. Maddalena, T. E. McKone and D. Mackay, Dependence of intake fraction on release location in a multimedia framework – A case study of four contaminants in North America, J. Ind. Ecol., 2004, 8, 89–102 CrossRef.
- C. Ng and N. von Goetz, The global food system as a transport pathway for hazardous chemicals: The missing link between emissions and exposure, Environ. Health Perspect., 2017, 125, 1–7 CrossRef PubMed.
- R. Gioia, L. Nizzetto, R. Lohmann, J. Dachs, C. Temme and K. C. Jones, Polychlorinated biphenyls (PCBs) in air and seawater of the Atlantic Ocean: Sources, trends and processes, Environ. Sci. Technol., 2008, 42, 1416–1422 CrossRef CAS PubMed.
- R. Gioia, S. Eckhardt, K. Breivik, F. M. Jaward, A. Prieto, L. Nizzetto and K. C. Jones, Evidence for major emissions of PCBs in the West African region, Environ. Sci. Technol., 2011, 45, 1349–1355 CrossRef CAS PubMed.
- K. Breivik, R. Gioia, P. Chakraborty, G. Zhang and K. C. Jones, Are reductions in industrial organic contaminants emissions in rich countries achieved partly by export of toxic wastes?, Environ. Sci. Technol., 2011, 45, 9154–9160 CrossRef CAS PubMed.
- C. Bogdal, E. Abad, M. Abalos, B. van Bavel, J. Hagberg, M. Scheringer and H. Fiedler, Worldwide distribution of persistent organic pollutants in air, including results of air monitoring by passive air sampling in five continents, Trends Anal. Chem., 2013, 46, 150–161 CrossRef CAS.
- R. Gioia, A. J. Akindele, S. A. Adebusoye, K. A. Asante, S. Tanabe, A. Buekens and A. J. Sasco, Polychlorinated biphenyls (PCBs) in Africa: a review of environmental levels, Environ. Sci. Pollut. Res., 2014, 21, 6278–6289 CrossRef CAS PubMed.
- O. P. Luzardo, L. D. Boada, C. Carranza, N. Ruiz-Suárez, L. A. Henríquez-Hernández, P. F. Valerón, M. Zumbado, M. Camacho and J. L. P. Arellano, Socioeconomic development as a determinant of the levels of organochlorine pesticides and PCBs in the inhabitants of Western and Central African countries, Sci. Total Environ., 2014, 497–498, 97–105 CrossRef CAS PubMed.
- N. Oreskes, K. Shrader-Frechette and K. Belitz, Verification, validation and confirmation of numerical models in the earth sciences, Science, 1994, 263, 641–646 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c8em00023a |
| This journal is © The Royal Society of Chemistry 2018 |