Refining the aggregate exposure pathway
Abstract
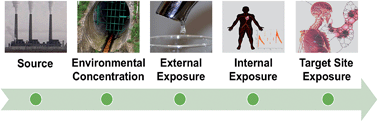
Advancements in measurement technologies and modeling capabilities continue to result in an abundance of exposure information, adding to that currently in existence. However, fragmentation within the exposure science community acts as an obstacle for realizing the vision set forth in the National Research Council's report on Exposure Science in the 21st century to consider exposures from source to dose, on multiple levels of integration, and to multiple stressors. The concept of an Aggregate Exposure Pathway (AEP) was proposed as a framework for organizing and integrating diverse exposure information that exists across numerous repositories and among multiple scientific fields. A workshop held in May 2016 followed introduction of the AEP concept, allowing members of the exposure science community to provide extensive evaluation and feedback regarding the framework's structure, key components, and applications. The current work briefly introduces topics discussed at the workshop and attempts to address key challenges involved in refining this framework. The resulting evolution in the AEP framework's features allows for facilitating acquisition, integration, organization, and transparent application and communication of exposure knowledge in a manner that is independent of its ultimate use, thereby enabling reuse of such information in many applications.
- This article is part of the themed collection: Modeling in Environmental Chemistry


 Please wait while we load your content...
Please wait while we load your content...