Quantifying variability in removal efficiencies of chemicals in activated sludge wastewater treatment plants – a meta-analytical approach†
Mélanie
Douziech
 *a,
Irene Rosique
Conesa
a,
Ana
Benítez-López
a,
Antonio
Franco
b,
Mark
Huijbregts
a and
Rosalie
van Zelm
a
*a,
Irene Rosique
Conesa
a,
Ana
Benítez-López
a,
Antonio
Franco
b,
Mark
Huijbregts
a and
Rosalie
van Zelm
a
aDepartment of Environmental Science, Institute for Water and Wetland Research, Radboud University Nijmegen, P.O. Box 9010, 6500 GL, Nijmegen, The Netherlands. E-mail: m.douziech@science.ru.nl
bSafety & Environmental Assurance Centre, Unilever, Colworth Science Park, Bedfordshire, MK441LQ, UK
First published on 2nd January 2018
Abstract
Large variations in removal efficiencies (REs) of chemicals have been reported for monitoring studies of activated sludge wastewater treatment plants (WWTPs). In this work, we conducted a meta-analysis on REs (1539 data points) for a set of 209 chemicals consisting of fragrances, surfactants, and pharmaceuticals in order to assess the drivers of the variability relating to inherent properties of the chemicals and operational parameters of activated sludge WWTPs. For a reduced dataset (n = 542), we developed a mixed-effect model (meta-regression) to explore the observed variability in REs for the chemicals using three chemical specific factors and four WWTP-related parameters. The overall removal efficiency of the set of chemicals was 82.1% (95% CI 75.2–87.1%, N = 1539). Our model accounted for 17% of the total variability in REs, while the process-based model SimpleTreat did not perform better than the average of the measured REs. We identified that, after accounting for other factors potentially influencing RE, readily biodegradable compounds were better removed than non-readily biodegradable ones. Further, we showed that REs increased with increasing sludge retention times (SRTs), especially for non-readily biodegradable compounds. Finally, our model highlighted a decrease in RE with increasing KOC. The counterintuitive relationship to KOC stresses the need for a better understanding of electrochemical interactions influencing the RE of ionisable chemicals. In addition, we highlighted the need to improve the modelling of chemicals that undergo deconjugation when predicting RE. Our meta-analysis represents a first step in better explaining the observed variability in measured REs of chemicals. It can be of particular help to prioritize the improvements required in existing process-based models to predict removal efficiencies of chemicals in WWTPs.
Environmental significanceThe amount of chemicals released into water bodies from consumer product use in developed countries mainly depends on the efficiency of wastewater treatment plants (WWTPs) to remove chemicals from wastewater. Current modelling approaches fail to explain the large variability observed in monitored removal efficiencies (REs). Applying a meta-analytical and meta-regression approach on a database of 1539 REs for surfactants, fragrances, and pharmaceuticals, we found empirical evidence that the sludge retention time of the WWTP, and biodegradation potential and partitioning to solids of the chemicals were important when modelling REs. Our model results also suggest that a better understanding of chemical-specific processes, like deconjugation and electrochemical interactions with sludge, are crucial for a better prediction of REs in WWTPs. |
1. Introduction
Activated sludge is a wide-spread technology for the treatment of wastewater and its constituent chemicals. The removal efficiency (RE) of specific chemicals in activated sludge wastewater treatment plants (WWTPs) is influenced by a wide number of factors. Luo et al. (2014) classified these factors as either internal or external. Internal factors are related to a chemical's properties, like the organic carbon–water partition coefficient (KOC) or its biodegradability. External factors encompass WWTP-related factors, such as the sludge retention time (SRT), the hydraulic retention time (HRT), or the water temperature.1–5 The influence of internal factors on the RE has been well studied. For example, a high sorption potential to organic carbon, indicated by a high KOC value, is a proxy for a high adsorption to sludge and high removal efficiencies in WWTPs.6–8 Furthermore, chemicals that are classified as readily biodegradable in stringent laboratory tests9,10 are typically assumed to be well removed during wastewater treatment.8,11 Such properties of chemicals are used in standard process-based models to estimate REs of chemicals in WWTPs.12 One example is the SimpleTreat model,13 which is widely-used in regulatory risk assessment to predict REs of chemicals in activated sludge WWTPs under standard operating conditions.14Attempts to describe the variability of measured removal efficiencies in process-based WWTP models like SimpleTreat have been made by using probabilistic modelling and Monte Carlo based uncertainty analysis.15,16 These studies, however, consider a probabilistic parameterization of a generic WWTP, not a plant-specific parameterization. Further, the variability of measured removal efficiencies has also been described by defining model parameters as function of operational conditions, for instance the biological degradation rate as a function of the SRT,17 although for a limited number of chemicals. Overall, the ability of standard process-based models to accurately predict the influence of specific parameters on REs across many chemicals and WWTPs remains to be proved. In a recent study on RE of pharmaceuticals, the predicted effluent concentrations from SimpleTreat were within a factor of 10 compared to actual measurements.18 Similarly, a factor of 10 was also reported for predicted effluent environmental concentrations, estimated with the STPWIN model from the EPI Suite programme, compared to measured values in Singer, et al.19 For fragrance ingredients, the variability in reported REs is large as well. For example, the reported RE of tonalide (a polycyclic musk) varied by a factor of six across a number of Dutch and American WWTPs.20–22 Some of this variability in REs may be explained by WWTP-related or so-called external factors. For example, higher REs for some chemicals have been observed for WWTPs with longer SRTs.23–25 Furthermore, parameters such as redox conditions, pH, or temperature are also presumed to influence the wastewater treatment process.2 Previously, studies investigated the influence of WWTP design parameters on the variability of REs. However, to our knowledge, the relationships unravelled are either specific to certain chemicals15 or WWTPs.26 Generalising these relationships to activated sludge WWTPs and more chemicals could be useful to provide guidance on how to include external factors in process-based models used in risk assessment and further potentially improve their prediction performance.
The aim of this research was to quantify the influence of both internal (chemical-related) and external (WWTP-related) factors on REs of a set of chemicals for which comprehensive and specific monitoring data on their removal in activated sludge WWTPs was available. The set of chemicals includes various types of surfactants, fragrance ingredients, and pharmaceuticals. We combined the RE data of each chemical in activated sludge WWTPs, with or without primary settler, with the chemical-specific and plant-specific parameters via a meta-analytical regression approach. To date, the only meta-analysis on REs of chemicals in WWTP compared the efficiency of five different sewage treatment technologies27 as opposed to one single technology such as activated sludge. We focused our assessment of variability on the various removal mechanisms occurring in activated sludge WWTPs using physico-chemical parameters together with operational WWTP parameters. In addition, we assessed the performance of the process-based WWTP model SimpleTreat in predicting the removal efficiencies of the chemicals.
2. Materials and methods
2.1. Literature review
We performed a literature review to identify studies of measured RE of surfactants, fragrances, and pharmaceuticals in activated sludge. For surfactants and fragrances, the search was carried out on ISI Web of Knowledge, PubMed, and Google Scholar without geographical and time constraints. For pharmaceuticals, the dataset gathered by Lautz, et al.18 was used as a basis and updated with an additional search in Web of Science, PubMed and Google Scholar. The search strings used for both searches are provided in the ESI (S1†).(1) The study had to refer to an activated sludge plant.
(2) The study had to report (i) the influent (before or after primary sedimentation) and effluent concentrations in wastewater (after the activated sludge treatment but before any tertiary treatment), or a combination of the RE and either the influent concentration or the effluent concentration of individual chemicals, and (ii) information on the WWTP's design.
(3) The study could report total or dissolved concentrations of influents and effluents. No transformation from dissolved to total concentration was applied due to lack of data.
(4) Studies reporting only the RE of a class of chemicals (e.g. anionic surfactants) were excluded.
(5) Degradation by-products were excluded from the analysis (e.g. alkylphenols and perfluorinated surfactants, and some pharmaceuticals like estrone, or estriol), as their generation term could not be properly captured in the modelling.
(6) Chemicals that might be generated through deconjugation of their metabolites were included in the model development. We assessed their potential influence on the model outcome via a sensitivity analysis (see Section 2.5).
(7) Compounds with multiple pKa in an environmentally relevant range (amphoters, zwitterions and multivalent ionic substances) were discarded. The highly pH dependent environmental behaviour of these compounds is not captured properly in the available models for environmental properties predictions.15,28
(8) Studies reporting influent concentrations lower than detection limits were discarded, as REs could not be calculated.
When WWTP specific parameters were reported as a range, the average of the minimum and maximum values was used for the modelling. For some operating parameters the following default values were assumed to reduce the number of missing values: presence of primary settler (13% of all data points), CSTR with OMR as secondary treatment (13% of all data points), presence of a final sedimentation tank (38% of all data points), and no tertiary treatment (6% of all data points). This standard design is also used in regulatory risk assessment frameworks (e.g. SimpleTreat). When information on the type of nutrient removal was available, it was used to refine the default secondary treatment type: a plug-flow reactor was assumed for BNR, and a CSTR for OMR.
Study-specific information was also recorded, namely the sampling period (winter, spring…), the sample type (composite or grab sample), the concentration measured (total or dissolved), the number of samples taken (sample size), the standard deviations (SD) of the measured concentrations, and whether influent and effluent concentrations were measured at the same time or offset by the HRT. Measured concentrations that account for the residence time in the WWTP (offset by the HRT) are expected to be more representative of REs. Not all parameters could be included in the final analysis due to low data availability and a detailed description of the required assumptions per single study is provided in the ESI, S2.†
In total, we selected 133 studies published between 1995 and 2017. Before any screening was performed, the raw database consisted of 2441 data points (or effect sizes, see Section 2.3.1). 1308 data points representing REs of the same chemical measured at different times but at the same WWTP were averaged (and SD calculated). Further screening, explained in the flowchart in the ESI, S1,† led to a final database consisting of records of 1539 REs for 209 chemicals, namely 34 fragrances, 21 surfactants, and 154 pharmaceuticals. The studies were distributed over 28 countries, with most studies conducted in Spain (24), the USA (15), and China (14). More information on the geographical coverage is provided in the ESI, S3† together with a list of all studies included.
2.2. Chemical properties
The PubChem, HERA, ECHA, DrugBank, ChemSpider, and eChemPortal websites were searched for experimental physico-chemical properties of the chemicals in the database.32–37 The properties recorded were: CAS number, molecular weight, pKa, octanol–water partitioning coefficient (KOW), organic carbon–water partitioning coefficient (KOC), Henry's law constant, biodegradability character (divided in five classes as explained below), and whether the component was a degradation by-product or not. The biodegradability of a chemical was determined from OECD test results, which classify a chemical into one of the following five classes, listed in decreasing order of biodegradability: (i) readily biodegradable; (ii) readily, but failing 10-d window; (iii) inherently biodegradable, fulfilling specific criteria; (iv) inherently biodegradable, not fulfilling specific criteria; and (v) not biodegradable.38 Biodegradability rates measured in published biodegradation studies were also considered. In these cases, the chemical was additionally classified in one of the five biodegradation classes using the indications from the European Commission Joint Research Centre.38 With this information, we defined two indicators of a chemical's biodegradability: (1) to which of the five classes the chemical belonged (biodegradability category), and (2) whether it was readily biodegradable (classes (i) and (ii)) or not readily biodegradable (classes (iii) to (v)).When no experimental data were available, the pKa and the KOW were estimated using the ACDLabs estimation programme.39 The dissociation equations provided by the estimation programme were used for a preliminary assessment of the chemical's class. Bases were classified as such, when the pKa was above 4, and acids whenever the pKa was smaller than 10. When no measured KOC was available, the pKa and KOW values were used to estimate KOC. Details of the equations applied are provided in the ESI, S2.† The ESI, S3† lists the chemicals considered together with their estimated or empirical physico-chemical properties. The speciation of the chemical was hereby assumed to occur at the optimal pH as derived by Franco and Trapp40 to ensure the best prediction possible. The Henry's law constant was calculated from the vapour pressure, molecular weight and solubility with the EPI Suite programme.41
In the recorded studies, concentrations were sometimes measured for surfactants with different carbon chain-length (i.e. LAS). In those cases, weighted averages of the chemical properties of the single components were computed either using the molecular weight, or, when available, the exact weight percentages of the single mixture's components. If one component in a mixture was not readily biodegradable, then the entire mixture was classed accordingly. Further explanation of the assumptions made to set up the chemicals' properties database are listed in the ESI, S2.†
2.3. Meta-analysis
Meta-analysis is a method to summarise studies using a common value, called the effect size, while accounting for the weights of the single studies (see Sections 2.3.1 and 2.3.2 for an explanation of the effect size and weights used in this study). Meta-regression models are a type of mixed-effects models that combine fixed and random effects to explain the observed variation in the effect size. Fixed effects, also called moderators, describe the effect size similarly to predictor variables in linear regression models. Random effects take into account additional heterogeneity, nested data, and temporal and spatial correlations.42 In our case, random effects were added to account for variability between chemicals, studies, and WWTPs and for the clustering nature of the data and to prevent that a few chemicals, studies and/or WWTPs, which are more often represented in the dataset, dominate the regression model results. A potential random effect parameter is the specific study the RE is taken from, as most likely not all parameters differentiating the studies are accounted for as fixed effects. Other random effect parameters are the WWTP and the chemical name. The chemical name is necessary to account for the fact that the same chemical repeated several times in the dataset is likely to have a similar response according to physicochemical properties that are unknown and not included in the fixed effects (pseudoreplication). The WWTP would capture the WWTP's specific characteristics and its exposition to similar environmental conditions, which would potentially affect the REs. Therefore, combining both fixed and random effects within one single mixed-effect model (meta-regression model) allows taking into account that the available studies are only a fraction of all studies potentially carried out, but still have common parameters influencing their outcome. Meta-analyses and meta-regression models were run using the metafor package43 in R v. 3.3.2.44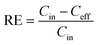 | (1) |
Because of the large spread in the REs reported (−249 to 1), this measure was not appropriate for the modelling.45 Instead, a response ratio (RR) for every WWTP-chemical combination was used as effect size (eqn (2)). Each RR was further weighted by the inverse of its sampling variance (eqn (3)) to assign more weight to studies with higher precision (i.e.: with smaller SD and larger sample sizes).45,46 Negative REs (n = 65) were not discarded from the analysis because our aim was to describe the broadest spectrum of observed REs possible. Potential explanations for these negative values would be the deconjugation of compounds, as for carbamazepine,47 or poor experimental design with e.g. simultaneous sampling of influent and effluent concentrations.
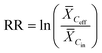 | (2) |
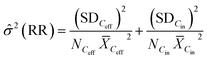 | (3) |
![[X with combining macron]](https://www.rsc.org/images/entities/i_char_0058_0304.gif) Ceff – mean of the effluent concentration,
Ceff – mean of the effluent concentration, ![[X with combining macron]](https://www.rsc.org/images/entities/i_char_0058_0304.gif) Cin – mean of the influent concentration,
Cin – mean of the influent concentration, ![[small sigma, Greek, circumflex]](https://www.rsc.org/images/entities/i_char_e111.gif) 2 – sampling variance, SDCeff – standard deviation of the effluent concentration, SDCin – standard deviation of the influent concentration, NCeff – number of samples for the effluent concentration, NCin – number of samples for the influent concentration.
2 – sampling variance, SDCeff – standard deviation of the effluent concentration, SDCin – standard deviation of the influent concentration, NCeff – number of samples for the effluent concentration, NCin – number of samples for the influent concentration.
We report our results as RE after back-transforming the RR using eqn (4).
| RE = 1 − exp(RR) | (4) |
Not all studies reported the sample size, nor did all report the standard deviations of the measured concentrations. We therefore used different imputation techniques to impute missing sample sizes and SDs using the package metagear.48 The random_N function was used to impute random sample sizes, whereas missing standard deviations were imputed using Bracken's method (impute_SD function), which uses the coefficient of variation of the entire dataset to impute standard deviations. Additionally, we imputed standard deviations using two other imputation techniques: the HotDeck and HotDeck Nearest Neighbour, and compared the results to our default approach (Bracken's method) (ESI, S8†). While the HotDeck method imputes SDs using all observed SDs, HotDeck Nearest Neighbour imputes only SDs from data with means of similar scale.49 In our case, we used the standard deviations from the three most similar RE values to the one with missing SD to impute the missing value.
The KOC, KOW, Henry's law constant, flow rate, HRT, and SRT were log-transformed to prevent the outliers from influencing the analysis too much.52 We also assessed multicollinearity between the moderators visually for the categorical variables and using Spearman correlation coefficients for the continuous ones. Moderators were assumed to be correlated for coefficients above 70%, and whenever clear differences in the box- and bar plots were visible. Given the large number of potential moderators, not all box- and barplots generated are shown. Instead, the reader can refer to the ESI, S4† for examples of those visual assessments. From each pair of correlated moderators, we kept the moderator that could influence the REs more directly and explained more heterogeneity in the REs (e.g.: the SRT was preferred over the secondary treatment type). This was assessed by exploring the strength and direction (positive or negative) of the relationship between RE and each single moderator using meta-regression models, with a special attention to Omnibus test results (Hedges and Pigott 2004). These models were fitted to a reduced database (542 data points), for which data for all the potential moderators were available to ensure comparability between the different models. In the end, seven moderators were retained for the model fitting (Table 1). More information on the screening process is provided in the ESI, S4.†
| Names | Justification | |
|---|---|---|
| T | log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sludge retention time sludge retention time |
Increased SRTs were found to increase the removal of chemicals25,26 |
| Type of biological nutrient removal | Proxy for the microbial communities in the secondary treatment tank: smaller REs were observed in OMR designs where neither nitrifying nor denitrifying bacteria are present25,53 | |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) flow rate flow rate |
Gives information about the size of the plant, the number of persons served, and the hydraulic retention time54 | |
| Share of domestic influent wastewater | Could influence the influent chemicals concentrations and the community of degrading organisms | |
| C | log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Henry constant Henry constant |
Describes the volatility of a substance: chemicals with high Henry constants will volatilise more and therefore be less in the water phase, thus leading to larger REs |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC KOC |
Determines the partition of a chemical between sludge and water: chemicals with higher KOC are likely to be less in the water than the sludge phase, thus leading to larger REs | |
| Readily biodegradable | Readily biodegradable substances are expected to be better removed during the wastewater treatment process25,55 | |
Readily biodegradable compounds are expected to degrade quickly in most environments including WWTPs10 and removal by sorption would be less relevant. In addition, the RE of readily biodegradable compounds is expected to be less sensitive to longer retention times in the WWTPs. In order to test these hypotheses, we included two interaction terms to the list of potential moderators, namely interaction between readily biodegradability and SRT and between readily biodegradability and KOC.
2.4. Model fitting
First, the optimal structure of the random effects was derived through comparison of models with the same full fixed-effects structure, meaning all seven moderators, and different random-effects.42 This was done with the reduced database of 542 data points to ensure comparability between the different models. Model selection was based on the Bayesian Information Criteria (BIC) value derived for models fitted with the restricted maximum likelihood method (REML) using the metafor package.43 The following parameters were considered to define the random effect structure of our models: the specific study in which the RE was reported, referred to as reference; the WWTP, using a unique plant code for each WWTP; the country the WWTP was located in; and the chemical name itself. This means that all these variables were assumed to potentially affect the RE by some aspects not captured in the fixed effect structures, e.g. country-specific climatic conditions, or additional physico-chemical properties. WWTP number was modelled as nested within a study, since each WWTP only occurred in one study.56 As a result, the WWTP code nested in the reference and the chemical name were kept as random effects (ESI, S5†). Further, the approach explained by Assink and Wibbelink57 was followed to quantify the contribution of each random effect to the total variance. This should allow inference about potentially missing explanatory variables. With this approach, we also quantified the contribution of the sampling variance, meaning within-study variance, to the total variance.In a second step, models with all possible fixed effects combinations and the optimal random effects structure were fitted to the reduced data set using the glmulti package and the maximum likelihood method.58 All candidate models were ranked using the BIC and the weight of each model was calculated. For the final model, fitted using REML method, we calculated the explained variance of the final model using Nakagawa's marginal R2 (ref. 59) (eqn(5)).
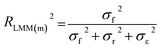 | (5) |
R LMM(m) 2, also called marginal R2, describes how much of the heterogeneity can be explained by the fixed effects compared to a total heterogeneity described by the variance in the fixed effects, random effects, and a mean sampling variance. The conditional R2, RLMM(c)2, describes the total heterogeneity explained by the model by summing up the variance of the fixed and random effects (σf2 + σr2) in the numerator of eqn (5). The compliance of the final model with the homogeneity, normality, and independence assumptions was assessed visually.42 Publication bias was also tested using Funnel plots and Egger tests comparing the sampling variance and the meta-analytic residuals.60,61
Finally, in addition to the meta-regression analysis, we estimated the mean weighted effect size across all studies. This value was back-transformed into an overall RE of the chemicals present in our database. The mean weighted RE was computed once for the total number of effect sizes available (1539 data points), and once for the reduced data set (542 data points).
2.5. Sensitivity to data quality
We scored each study according to quality and ran sensitivity analyses to assess the robustness of our results when low quality studies were removed. The quality index was based on four criteria as described in Table 2. Per criterion, the study was either ranked as “good”, “moderate”, or “poor”. It appeared that only six out of the 133 studies ranked good for all quality criteria tested. As a result, conducting a separate meta-analysis on them was not possible. The influence of data quality on our analysis was therefore tested for two different scenarios:| Good | Moderate | Poor | |
|---|---|---|---|
| a The following chemicals were potentially formed by deconjugation of their metabolites: estradiol, fluoxetine, naproxen, diclofenac, ketoprofen, diazepam, prazepam, temazepam, carbamazepine, sulfamethoxazole, erythromycin, codeine and amitriptyline. | |||
| Criterion 1 – measurement relevant to total concentration | Total concentrations reported | Dissolved concentrations for chemicals with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC < 3 KOC < 3 |
Dissolved concentrations for chemicals with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC > 3 KOC > 3 |
| Criterion 2 – type of sample | Flow proportional composite sample | Combined grab samples, volume-proportional or time-proportional composite sample | Single grab samples |
| Criterion 3 – experimental values available for KOC, readily biodegradability | Both values are experimental | One property only was experimental | No property experimental |
| Criterion 4 – potentially formed by deconjugation of metabolitesa | No | No | Yes |
(1) including all studies, which is the default scenario, and
(2) including studies scoring either “good” or “moderate” in all criteria.
2.6. SimpleTreat
We applied SimpleTreat 4.0 to the data points in our database and assessed its performance. We used the Henry constant, KOC, and biodegradation rate as physico-chemical properties' input. Where available, experimental biodegradation rates were used directly. Whenever only a classification according to the OECD test results was available, rates were assigned according to SimpleTreat's classification scheme.62 The sludge loading rate was derived from the study's specific SRT according to the indications in Struijs.62 Further, the presence of primary settler was adapted per study given the information provided, and where available the per capita flow rate was also used as input parameter. The performance of SimpleTreat was then assessed using eqn (6), thus basing on the comparison of the response ratios (RRs).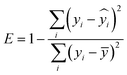 | (6) |
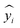 – estimated RR, ȳ – average of the measured RRs.
– estimated RR, ȳ – average of the measured RRs.
3. Results
3.1. Univariate moderator influence
The mean weighted RE across studies of the entire database was 82.1% (95% CI = 75.2–87.1%, N = 1539). For the reduced dataset (N = 542), that was used in the meta-regression, the mean weighted RE was 64.9% (95% CI: 49.4–75.7%). The implications of varying data quality on this mean weighted RE are presented in Section 3.3 and discussed in Section 4.2.When looking at single mixed-effect models we found that the readily biodegradability of a substance and the SRT of the plant had a significant influence on the RE (Qm = 24.6, P = 7.2 × 10−7 and Qm = 4.5 and P = 0.03, respectively) (ESI, S4†). The RE increased with increased SRTs. Further, as expected, readily biodegradable chemicals were on average better removed than non-readily biodegradable ones (91% with 95% CI = 38.5–70.2% vs. 57% with 95% CI = 83.5–95.6%).
3.2. Meta-regression model
The final averaged model according to BIC included the chemical's readily biodegradability, the SRT, their interaction, as well as the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC and its interaction to the chemical's readily biodegradability as moderators. The readily biodegradability, the log
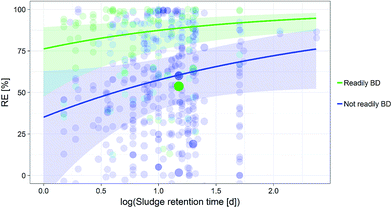
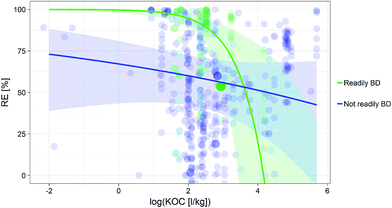
KOC and its interaction to the chemical's readily biodegradability as moderators. The readily biodegradability, the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC and its interaction to the chemical's readily biodegradability were hereby influencing the RE most according to the Omnibus test result (ESI, S6†). Overall, REs were higher for readily biodegradable compounds (Fig. 1). Furthermore, we found that RE increased with increasing SRT, particularly for non-readily biodegradable compounds (Fig. 2).
KOC and its interaction to the chemical's readily biodegradability were hereby influencing the RE most according to the Omnibus test result (ESI, S6†). Overall, REs were higher for readily biodegradable compounds (Fig. 1). Furthermore, we found that RE increased with increasing SRT, particularly for non-readily biodegradable compounds (Fig. 2).
Further, it appears from Fig. 2 that the confidence interval of the relationship between the KOC and readily biodegradable compounds is widening a lot for log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC values above 2.5.
KOC values above 2.5.
The fixed effects explained 17% of the observed heterogeneity (marginal R2), while fixed and random effects together account for 96% of the observed heterogeneity (conditional R2). From all random effects included, the chemical name explained most of the variance accounted for by the random effects (52.4%), while the reference accounted for 40.9%, and the plant code for only 6.5% (ESI, S5†). The final model met homogeneity, normality and independence assumptions, and was not influenced by publication bias (ESI, S7†).
3.3. Sensitivity to data quality
Firstly, we saw that the different imputation strategies applied to estimate missing standard deviation of the single measured concentrations did not substantially influence the mean weighted RE (ESI, S8†).Secondly, for the entire and reduced databases (N = 1539 and 542, respectively), about 35% of the data had moderate or good scores for all categories. The mean weighted removal efficiencies of these data subsets were higher than for the raw datasets. Excluding the data scoring poor in any criteria, increased the mean weighted RE of the reduced dataset from 64.9% (95% CI = 49.4–75.7%, N = 542) to 71.6% (95% CI = 44.9–85.3%, N = 193) and the one of the entire database from 82.1% (95% CI = 75.2–87.1%, N = 1539) to 87.2% (95% CI = 78.5–92.4%, N = 522).
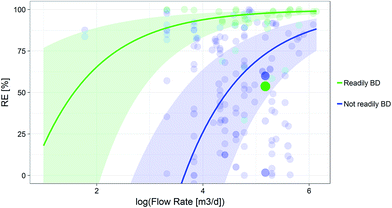
Finally, the mixed effect model fitted to the reduced dataset with data points of moderate or good scores in all categories (N = 193) retained the same moderators as the final model plus the flow rate to explain the observed heterogeneities. The latter significantly influenced the RE according to the Omnibus test (Qm = 65.2, P = 6.8 × 10−16, S7†). Fig. 3 shows the increase in RE observed with increasing flow rate. The relationships between the RE and the SRT and KOC are very similar to the ones presented in Fig. 1 and 2 (ESI, S7†).
The fixed effects explained 32% of the observed heterogeneity (marginal R2), while fixed and random effects together account for 98% of the observed heterogeneity (conditional R2). The chemical name explained 19.1% of the variance accounted for by the random effects, while the reference accounted for 68.4%, and the plant code for 12.4%.
3.4. SimpleTreat
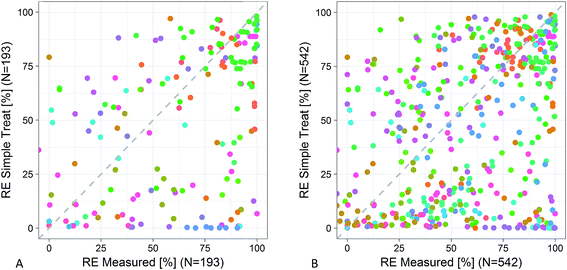
SimpleTreat showed a coefficient of efficiency of −0.04 for the reduced dataset (N = 542), thus meaning that the estimates are not better predictors than the average value of the measured data. This value rose to 0.14 when studies of only good and moderate quality criteria were included. Overall the fit was however rather poor as reflected in Fig. 4.4. Discussion
4.1. Heterogeneity in removal efficiencies
The RE computed for the entire dataset was relatively high (82.1%) compared to previous, more chemical class specific studies, reporting the poor performances of activated sludge WWTPs in removing pharmaceuticals.63–65 Although this high RE may be due to the inclusion in our database of repeated measurements of rather well removed chemicals like fragrances, or ibuprofen, or diclofenac, we accounted for pseudoreplication by including chemical name as random effect in our models. Thus, this should not be affecting our estimate. Further data quality should also not affect the mean weighted removal efficiency derived, which is demonstrated by the similar mean weighted RE derived for the dataset excluding data scoring poor in any of the quality criterion (87.2%). The main driving factor for the difference observed is the number of different chemicals for which high and low REs are reported. In fact, for the entire database, 111 different chemicals are reported to have REs above 90% while only 32 are found in the reduced dataset. The 82.1% weighted removal efficiency is therefore a representative RE estimate of all chemicals included in the total database.Overall, our findings highlight the importance of SRT in determining removal efficiency especially for the non-readily biodegradable chemicals in the dataset. Further, we identified biotransformation/degradation as the main removal mechanism for many chemicals, similarly to the outcomes of other studies.25,55 An increase in RE of non-readily biodegradable chemicals observed for higher SRTs has also been reported2 such as in the case of certain pharmaceuticals which have been shown to degrade under aerobic nitrifying conditions.25 Clara, et al.26 also support the use of longer SRTs to increase the removal of pharmaceuticals. We should stress that the importance of the SRT on the RE might not only be related to changes in the microbial community. In fact, we found that the SRT and the HRT were correlated (Spearman correlation around 50%), and thus, a higher SRT could be interpreted as a surrogate of a high HRT, and therefore indicate longer residence time of a chemical in the WWTP and, as a result, higher degradation. The influence of the flow rate on REs of the reduced dataset could be interpreted as an indicator of the treatment capacity of the WWTPs.
Our model also identified a decrease in RE with increasing KOC value. Interestingly, in process-based models predicting the removal of chemicals in activated sludge WWTPs, higher KOC are expected to increase a chemical's removal. The counterintuitive relationship we identified is likely driven by the low REs identified for compounds with high KOC values. Compounds with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC values above 3.5 are known to have strong adsorption to organic material and this especially for WWTP with high fraction of organic carbon and suspended solid concentrations.66 This is the case for the readily biodegradable compounds fluoxetine and roxithromycin (log
KOC values above 3.5 are known to have strong adsorption to organic material and this especially for WWTP with high fraction of organic carbon and suspended solid concentrations.66 This is the case for the readily biodegradable compounds fluoxetine and roxithromycin (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC = 4.2 and 2.9, respectively) and the non-readily biodegradable compounds paroxetine and sertraline (log
KOC = 4.2 and 2.9, respectively) and the non-readily biodegradable compounds paroxetine and sertraline (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC > 4). Possible non-mutually exclusive explanations for this relationship are: (1) the existence of electrical interactions of pharmaceuticals to sludge;67–69 (2) the existence of other processes affecting removal and not captured by our moderators (e.g. deconjugation);15 (3) the large spread sometimes observed in the experimentally derived KOC values;70 and (4) the lack of experimental data as a whole, especially for measured REs of readily biodegradable compounds with log
KOC > 4). Possible non-mutually exclusive explanations for this relationship are: (1) the existence of electrical interactions of pharmaceuticals to sludge;67–69 (2) the existence of other processes affecting removal and not captured by our moderators (e.g. deconjugation);15 (3) the large spread sometimes observed in the experimentally derived KOC values;70 and (4) the lack of experimental data as a whole, especially for measured REs of readily biodegradable compounds with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOC values above 2.5 and reflected in the wider confidence interval computed for this relationship. In fact, Tolls71 suggests to consider not only sorption to organic matter, but also surfactant adsorption to mineral constituents, ion exchange, and reactions to account for all potential mechanisms influencing the sorption of cations to sludge. Similarly, Berthod, et al.72 observed that charge–charge Coulomb interactions could be as effective as hydrophobic interactions to explain the sorption of cations to sludge.
KOC values above 2.5 and reflected in the wider confidence interval computed for this relationship. In fact, Tolls71 suggests to consider not only sorption to organic matter, but also surfactant adsorption to mineral constituents, ion exchange, and reactions to account for all potential mechanisms influencing the sorption of cations to sludge. Similarly, Berthod, et al.72 observed that charge–charge Coulomb interactions could be as effective as hydrophobic interactions to explain the sorption of cations to sludge.
The relation of other parameters to the RE of chemicals in activated sludge WWTPs was explored as well. The Henry constant was weakly negatively related to RE, probably because pharmaceuticals, which represented most of the chemicals in our database (N = 88%), have low volatility.
The importance of the random factors in explaining the heterogeneity further indicates that removal is influenced by other parameters, unspecified in the model. For the dataset where no data quality screening was performed, the importance of the chemical name as random factor highlights missing moderators specific to the chemical to explain the heterogeneity in removal. In fact, further parameters such as the surface area, the polarity, or the charge of the molecule could affect its interactions with other molecules and hence also its removal from wastewater.67,69,73,74 Fitting our model to a subset of our data following stricter quality criteria, showed, however, that the random effects of WWTP-specific properties played a more important role than physico-chemical properties compared to the model with all data points included. Note that nearly half of the data points excluded in the restricted model refer to chemicals that may be partly formed during deconjugation of their metabolites. The change in importance of the random effect factors in the reduced compared to the full model points towards the potential importance of a thorough modelling of chemical-specific deconjugation processes in activated sludge WWTPs.
4.2. Model performance
Considering subsets of higher quality data only slightly improved the mean weighted RE of the dataset compared to the raw one. Further, the use of different imputation strategies did also not greatly affect the mean weighted RE derived. These observations support the choices made and the wider applicability of our approach.Still, the large difference between the marginal R2 of both models (17% and 32%) and the conditional R2 (96% and 98%) shows that the major part of the heterogeneity is not explained by our choice of fixed moderators, although these have been identified in many previous studies as important factors affecting RE.25,26,53 Here, the variation in the moderator's identified for a different subset of chemicals helped us draw conclusions on potential processes missing in our model (e.g. deconjugation). We therefore argue that future research should be aimed at better incorporating complex removal processes, like deconjugation, into simple process-based models, like SimpleTreat. In fact, chemical-specific deconjugation has also already been identified in other studies as influencing the removal of chemicals in activated sludge WWTPs.15 Further, the counterintuitive relationship of the RE to the KOC stresses the need for a better understanding of the electrochemical interactions of pharmaceuticals to activated sludge (see also Kummerer,68 Zhao, et al.,67 Tolls71).
The random effect part of WWTP specific parameters was also shown to be important. Several studies have reported the influence of nitrifying conditions, pH, and temperature on the REs,2,25,54 but the availability of these parameters was limited across the studies included in the meta-analysis. For other parameters, e.g. the presence of primary settler, the predominance of one design type in the investigated WWTP prevented an analysis of their influence on the results. Another aspect our model did not account for is temporal variability. Influent loads of chemicals can vary throughout the day and the year.75,76 An increased load might potentially affect the performance of the WWTP in removing the chemicals, as it could influence the bacterial community.77 Further, seasonal variability induced by changes in temperature, in turn impacting the performance of the bacteria in the system, might also reduce the performance of activated sludge WWTPs.78 The reported information was however not sufficient to properly assess this aspect. Additional monitoring studies would be necessary to thoroughly understand their influence on REs.
Finding a way to include these parameters into process-based models like SimpleTreat appears even more important given the poor agreement between measured and predicted REs by SimpleTreat. Besides the explanations provided above concerning the deconjugation processes and electrochemical interactions, a better differentiation of the redox conditions present in the WWTP could also improve SimpleTreat's predictions.
4.3. Limitations and uncertainties
Explaining the heterogeneities observed in the monitored removal of chemicals from activated sludge WWTPs was complicated by (1) the complexity of modelling chemical's behaviour in the environment, (2) the lack of experimental data in measured REs as well as physico-chemical properties, (3) the large variability in the experimental settings, (4) the lack of uniformity in the way measured results were generated and reported, and (5) the uncertainty in the reported concentrations. The first point is typified by perfluorinated surfactants that were excluded on the basis of their complex chemistry and the difficulty to identify the parent chemical. Excluding perfluorinated surfactants reduced the number of data points for surfactants by 90%. However, complex chemistry, the lack of measured physico-chemical properties, and the use of screening biodegradability test results also limit the application of process-based models like SimpleTreat.Differences in the way environmental concentrations are measured and reported justified the use of mixed-effect models. These models can account for such between-study variability, among other sources of variation (different WWTP, different chemicals, etc.), whereas conventional regression models cannot. However, using a mixed-effect model does not guarantee to account for all variations in study design. For example, some studies reported only dissolved concentrations, while others measured total concentrations. A possible solution would have been to estimate total concentration by adding the fraction of chemicals bound to suspended solids to the dissolved concentration. However, this approach would have implied assuming an influent suspended solids concentration and an organic carbon fraction, besides using an often estimated KOC. Given the uncertainty induced by this procedure and the potential bias to the KOC, we decided to use the data as reported in the papers. The overall RE reported here might therefore be underestimated, as the influent concentration of chemical might be underestimated.
Further, the lack of uniformity in the way monitoring studies reported their results reduced the choice of potential moderators. Very few studies, for example, reported concentrations of suspended solids, water temperatures, or pH values (on average less than 20%). Also, many studies could not be considered because only averaged concentrations over different plants were reported, or no details on the methodology and/or the WWTP were available.
Finally, assuming half of the LOD or LOQ whenever concentrations were below these thresholds also introduces additional uncertainty in the derived REs, which was not quantified in our study.
5. Conclusion
Large variability exists in monitored REs between types of chemicals but also for a single chemical depending on a range of factors other than chemical-specific properties and this is not fully accounted for in process-based models using mainly physico-chemical properties to predict REs. Using mixed-effect models, we showed that WWTP design parameters could help to explain some of this heterogeneity. We highlighted the importance of the SRT as well as the flow rate. Additional research to identify the exact relationship between the SRT and the REs could be beneficial for process-based models like SimpleTreat, which implement a categorical relationship between the SRT and the sludge loading rate.More work is further necessary to thoroughly identify all potential influencing factors. In fact, there are still examples of reported concentrations for which the design parameters fail to explain observed differences. In these cases, our analysis, in line with other recent developments,15 pointed out that a better modelling of deconjugation processes and electrochemical interactions of chemicals with sludge could improve RE predictions.
Overall, a recommendation for future monitoring studies would be to measure of characterise the WWTP and its performance with more parameters. Special attention should be given to the SRT as this is an important factor in determining the microbial community which in turn is responsible for the biodegradation and removal of chemicals. Extensive studies are however missing, thus calling for more work in this area.
Conflicts of interest
There are no conflicts of interest to declare.Acknowledgements
We would like to thank Manfred Clara, Eva Pocurull, Marion Letzel, Athanasios Stasinakis, Michael Gardner, Shirley Anne Smyth, Jakob Strand, Riccardo Gori, and Hideshige Takada for sharing the raw data of their study and/or answering important questions about the methodology they applied. The help of Michael Cunningham in choosing the relevant WWTP operating parameters was also very precious. Further, we would like to acknowledge Leonie Lautz for sharing her database and for the details she gave about its creation. The explanations of Wolfgang Viechtbauer on best practices in meta-analysis helped to improve the understanding of some concepts presented here. We would further like to thank David Sheffield for his help on specific statistical concepts. Finally, we also thank Rik Oldenkamp for sharing his data on the biodegradability of pharmaceuticals and his implementation of SimpleTreat in R. This project was funded by the European Union's Horizon 2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement No 641459 (RELIEF).References
- U. Klaschka, P. C. von der Ohe, A. Bschorer, S. Krezmer, M. Sengl and M. Letzel, Occurrences and potential risks of 16 fragrances in five German sewage treatment plants and their receiving waters, Environ. Sci. Pollut. Res. Int., 2013, 20(4), 2456–2471 CrossRef CAS PubMed.
- Y. Luo, W. Guo, H. H. Ngo, L. D. Nghiem, F. I. Hai, J. Zhang, S. Liang and X. C. Wang, A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment, Sci. Total Environ., 2014, 473–474, 619–641 CrossRef CAS PubMed.
- A. Joss, E. Keller, A. C. Alder, A. Gobel, C. S. McArdell, T. Ternes and H. Siegrist, Removal of pharmaceuticals and fragrances in biological wastewater treatment, Water Res., 2005, 39(14), 3139–3152 CrossRef CAS PubMed.
- M. Gros, M. Petrović and D. Barceló, Wastewater treatment plants as a pathway for aquatic contamination by pharmaceuticals in the Ebro river basin (Northeast Spain), Environ. Toxicol. Chem., 2007, 26(8), 1553–1562 CrossRef CAS PubMed.
- Q. Sun, M. Lv, A. Hu, X. Yang and C.-P. Yu, Seasonal variation in the occurrence and removal of pharmaceuticals and personal care products in a wastewater treatment plant in Xiamen, China, J. Hazard. Mater., 2014, 277, 69–75 CrossRef CAS PubMed.
- M. Clara, O. Gans, G. Windhofer, U. Krenn, W. Hartl, K. Braun, S. Scharf and C. Scheffknecht, Occurrence of polycyclic musks in wastewater and receiving water bodies and fate during wastewater treatment, Chemosphere, 2011, 82(8), 1116–1123 CrossRef CAS PubMed.
- T. Ternes, A. Joss and H. Siegrist, Scrutinizing pharmaceuticals and personal care products in wastewater treatment, Environ. Sci. Technol., 2004, 393–399 Search PubMed.
- R. Salgado, R. Marques, J. P. Noronha, G. Carvalho, A. Oehmen and M. A. Reis, Assessing the removal of pharmaceuticals and personal care products in a full-scale activated sludge plant, Environ. Sci. Pollut. Res. Int., 2012, 19(5), 1818–1827 CrossRef CAS PubMed.
- European Chemicals Agency REACH legislation, https://echa.europa.eu/regulations/reach/legislation, accessed 10.
- DOFO Chemicals, Guideline for testing of chemicals, OECD, 2005 Search PubMed.
- C. G. Daughton and T. A. Ternes, Pharmaceuticals and Personal Care Products in the Environment: Agents of Subtle Change?, Environ. Health, 1999, 107(6), 907–938 CAS.
- M. Pomies, J. M. Choubert, C. Wisniewski and M. Coquery, Modelling of micropollutant removal in biological wastewater treatments: a review, Sci. Total Environ., 2013, 443, 733–748 CrossRef CAS PubMed.
- J. Struijs, SimpleTreat 3.0: a model to predict the distribution and elimination of chemicals by sewage treatment plants, 719101025, National Institute of public health and the environment, Bilthoven, The Netherlands, 1996 Search PubMed.
- J. Struijs, D. van de Meent, D. Schowanek, H. Buchholz, R. Patoux, T. Wolf, T. Austin, J. Tolls, K. van Leeuwen and M. Galay-Burgos, Adapting SimpleTreat for simulating behaviour of chemical substances during industrial sewage treatment, Chemosphere, 2016, 159, 619–627 CrossRef CAS PubMed.
- F. Polesel, H. R. Andersen, S. Trapp and B. G. Plosz, Removal of Antibiotics in Biological Wastewater Treatment Systems-A Critical Assessment Using the Activated Sludge Modeling Framework for Xenobiotics (ASM-X), Environ. Sci. Technol., 2016, 50(19), 10316–10334 CrossRef CAS PubMed.
- A. Franco, J. Struijs, T. Gouin and O. R. Price, Evolution of the sewage treatment plant model SimpleTreat: use of realistic biodegradability tests in probabilistic model simulations, Integr. Environ. Assess. Manage., 2013, 9(4), 569–579 CrossRef CAS PubMed.
- B. G. Plosz, K. H. Langford and K. V. Thomas, An activated sludge modeling framework for xenobiotic trace chemicals (ASM-X): assessment of diclofenac and carbamazepine, Biotechnol. Bioeng., 2012, 109(11), 2757–2769 CrossRef CAS PubMed.
- L. S. Lautz, J. Struijs, T. M. Nolte, A. M. Breure, E. van der Grinten, D. van de Meent and R. van Zelm, Evaluation of SimpleTreat 4.0: Simulations of pharmaceutical removal in wastewater treatment plant facilities, Chemosphere, 2017, 168, 870–876 CrossRef CAS PubMed.
- H. P. Singer, A. E. Wössner, C. S. McArdell and K. Fenner, Rapid screening for exposure to “non-target” pharmaceuticals from wastewater effluents by combining HRMS-based suspect screening and exposure modeling, Environ. Sci. Technol., 2016, 50, 6698–6707 CrossRef CAS PubMed.
- E. Artola-Garciano, I. Borkent, J. L. M. Hermens and W. H. J. Vaes, Removal of two polycyclic musks in sewage treatment plants: freely dissolved and total concentrations, Environ. Sci. Technol., 2003, 37, 3111–3116 CrossRef.
- J. L. Reiner, J. D. Berset and K. Kannan, Mass flow of polycyclic musks in two wastewater treatment plants, Arch. Environ. Contam. Toxicol., 2007, 52(4), 451–457 CrossRef CAS PubMed.
- S. L. Simonich, W. M. Begley, G. Debaere and W. S. Eckhoff, Trace analysis of fragrance material in wastewater and treated wastewater, Environ. Sci. Technol., 2000, 34, 959–965 CrossRef CAS.
- A. Wick, G. Fink, A. Joss, H. Siegrist and T. A. Ternes, Fate of beta blockers and psycho-active drugs in conventional wastewater treatment, Water Res., 2009, 43(4), 1060–1074 CrossRef CAS PubMed.
- M. Clara, B. Strenn, O. Gans, E. Martinez, N. Kreuzinger and H. Kroiss, Removal of selected pharmaceuticals, fragrances and endocrine disrupting compounds in a membrane bioreactor and conventional wastewater treatment plants, Water Res., 2005, 39(19), 4797–4807 CrossRef CAS PubMed.
- S. Suarez, J. M. Lema and F. Omil, Removal of pharmaceutical and personal care products (PPCPs) under nitrifying and denitrifying conditions, Water Res., 2010, 44(10), 3214–3224 CrossRef CAS PubMed.
- M. Clara, N. Kreuzinger, B. Strenn, O. Gans and H. Kroiss, The solids retention time-a suitable design parameter to evaluate the capacity of wastewater treatment plants to remove micropollutants, Water Res., 2005, 39(1), 97–106 CrossRef CAS PubMed.
- S. D. Melvin and F. D. Leusch, Removal of trace organic contaminants from domestic wastewater: A meta-analysis comparison of sewage treatment technologies, Environ. Int., 2016, 92–93, 183–188 CrossRef CAS PubMed.
- R. Oldenkamp, M. A. Huijbregts, A. Hollander, A. Versporten, H. Goossens and A. M. Ragas, Spatially explicit prioritization of human antibiotics and antineoplastics in Europe, Environ. Int., 2013, 51, 13–26 CrossRef CAS PubMed.
- GetData Graph Digitizer, Digitizing software. Digitize scanned graphs and get original (x,y) data, 2017 Search PubMed.
- E. Sinclair and K. Kannan, Mass loading and fate of perfluoroalkyl surfactants in wastewater treatment plants, Environ. Sci. Technol., 2006, 40(5), 1408–1414 CrossRef CAS PubMed.
- R. Guo, W. J. Sim, E. S. Lee, J. H. Lee and J. E. Oh, Evaluation of the fate of perfluoroalkyl compounds in wastewater treatment plants, Water Res., 2010, 44(11), 3476–3486 CrossRef CAS PubMed.
- European Chemical Agency, Search for Chemicals, 2015 Search PubMed.
- DrugBank Seproxetine, https://www.drugbank.ca/drugs/DB06731, accessed 06.
- National Center for biotechnology information PubChem,https://pubchem.ncbi.nlm.nih.gov/, accessed 07.
- HERA Human and environmental risk assessment on ingredients of household cleaning products, http://www.heraproject.com/, accessed 07.
- Royal Society of Chemistry ChemSpider, Search and share chemistry, http://www.chemspider.com/, accessed 07.
- OECD The Global portal to information on chemical substances, https://www.echemportal.org/echemportal/index.action.
- European Commission Joint Research Centre, Technical Guidance Document on Risk Assessment Part II, Institute for Health and Consumer Protection, 2003 Search PubMed.
- ACD/Labs ACD/Labs, http://www.acdlabs.com/, accessed 06.
- A. Franco and S. Trapp, Estimation of the soil-water partition coefficient normalized to organic carbon for ionizable organic chemicals, Environ. Toxicol. Chem., 2008, 27(10), 1995–2004 CrossRef CAS PubMed.
- EPA 5, Estimating Physical/Chemical and Environmental Fate Properties with EPI Suite, U.S. Environmental Protection Agency, 2015 Search PubMed.
- A. Zuur, E. Ieno, N. Walker, A. Saveliev and G. Smith, Mixed effects models and extensions in ecology with R, Springer, New York, 2009 Search PubMed.
- W. Viechtbauer, Conducting meta-analysis in R with the metafor package, J. Stat. Softw., 2010, 36(3), 1–48 Search PubMed.
- R Core Team, R: A language and environment for statistical computing, R Foundation for Statistical Computing, Vienna, Austria, 2016 Search PubMed.
- M. Borenstein, L. V. Hedges, J. P. Higgins and H. R. Rothstein, Introduction to Meta-Analysis, John Wiley & Sons, Ltd., 2009 Search PubMed.
- M. J. Lajeunesse, On the meta-analysis of response ratios for studies with correlated and multi-group designs, Ecology, 2011, 92(11), 2049–2055 CrossRef.
- K. H. Langford and K. V. Thomas, Determination of pharmaceutical compounds in hospital effluents and their contribution to wastewater treatment works, Environ. Int., 2009, 35(5), 766–770 CrossRef CAS PubMed.
- M. J. Lajeunesse, Comprehensive Research Synthesis Tools for systematic reviews and meta-analysis, 2015 Search PubMed.
- D. B. Rubin and N. Schencker, Multiple imputation in health-care databases: an overview and some applications, Stat. Med., 1991, 10, 585–598 CrossRef CAS PubMed.
- B. Petrie, E. J. McAdam, J. N. Lester and E. Cartmell, Assessing potential modifications to the activated sludge process to improve simultaneous removal of a diverse range of micropollutants, Water Res., 2014, 62, 180–192 CrossRef CAS PubMed.
- S. L. Simonich, T. W. Federle, W. S. Eckhoff, A. Rottiers, S. Webb, D. Sabaliunas and W. de Wolf, Removal of fragrance material during U.S. and European wastewater treatment, Environ. Sci. Technol., 2002, 36, 2839–2847 CrossRef CAS PubMed.
- A. F. Zuur, E. N. Ieno and C. S. Elphick, A protocol for data exploration to avoid common statistical problems, Methods Ecol. Evol., 2010, 1(1), 3–14 CrossRef.
- E. J. McAdam, J. P. Bagnall, Y. K. Koh, T. Y. Chiu, S. Pollard, M. D. Scrimshaw, J. N. Lester and E. Cartmell, Removal of steroid estrogens in carbonaceous and nitrifying activated sludge processes, Chemosphere, 2010, 81(1), 1–6 CrossRef CAS PubMed.
- T. Deblonde, C. Cossu-Leguille and P. Hartemann, Emerging pollutants in wastewater: a review of the literature, Int. J. Hyg. Environ. Health, 2011, 214(6), 442–448 CrossRef CAS PubMed.
- H. R. Andersen, M. Hansen, J. Kjolholt, F. Stuer-Lauridsen, T. Ternes and B. Halling-Sorensen, Assessment of the importance of sorption for steroid estrogens removal during activated sludge treatment, Chemosphere, 2005, 61(1), 139–146 CrossRef CAS PubMed.
- H. Schielzeth, S. Nakagawa and R. Freckleton, Nested by design: model fitting and interpretation in a mixed model era, Methods Ecol. Evol., 2013, 4(1), 14–24 CrossRef.
- M. Assink and C. J. M. Wibbelink, Fitting three-level meta-analytic models in R: A step-by-step tutorial, Quant. Methods Psychol., 2016, 12(3), 154–174 CrossRef.
- V. Calcagno and C. de Mazancourt, glmulti: An R package for easy automated model selection with (generalized) linear models, J. Stat. Softw., 2010, 34(12), 1–28 Search PubMed.
- S. Nakagawa, H. Schielzeth and R. B. O'Hara, A general and simple method for obtaining R2 from generalized linear mixed-effects models, Methods Ecol. Evol., 2013, 4(2), 133–142 CrossRef.
- M. Egger, G. D. Smith, M. Schneider and C. Minder, Bias in meta-analysis detected by a simple, graphical test, BMJ, 1997, 315, 629–634 CrossRef CAS PubMed.
- S. Nakagawa and E. S. A. Santos, Methodological issues and advances in biological meta-analysis, Evol. Ecol., 2012, 26(5), 1253–1274 CrossRef.
- J. Struijs, SimpleTreat 4.0: a model to predict the fate and emission of chemicals in wastewater treatment plants, 2014 Search PubMed.
- H. Q. Liu, J. C. Lam, W. W. Li, H. Q. Yu and P. K. Lam, Spatial distribution and removal performance of pharmaceuticals in municipal wastewater treatment plants in China, Sci. Total Environ., 2017, 586, 1162–1169 CrossRef CAS PubMed.
- L. Lishman, S. A. Smyth, K. Sarafin, S. Kleywegt, J. Toito, T. Peart, B. Lee, M. Servos, M. Beland and P. Seto, Occurrence and reductions of pharmaceuticals and personal care products and estrogens by municipal wastewater treatment plants in Ontario, Canada, Sci. Total Environ., 2006, 367(2–3), 544–558 CrossRef CAS PubMed.
- N. Lindqvist, T. Tuhkanen and L. Kronberg, Occurrence of acidic pharmaceuticals in raw and treated sewages and in receiving waters, Water Res., 2005, 39(11), 2219–2228 CrossRef CAS PubMed.
- EPA, EPI Suite – Estimation Program Interface, https://www.epa.gov/tsca-screening-tools/epi-suitetm-estimation-program-interface, accessed 11, 2017.
- L. Zhao, Y. Ji, J. Yao, S. Long, D. Li and Y. Yang, Quantifying the fate and risk assessment of different antibiotics during wastewater treatment using a Monte Carlo simulation, J. Cleaner Prod., 2017, 626–631 CrossRef CAS.
- K. Kummerer, Antibiotics in the aquatic environment–a review–part I, Chemosphere, 2009, 75(4), 417–434 CrossRef PubMed.
- E. Torresi, F. Polesel, K. Bester, M. Christensson, B. F. Smets, S. Trapp, H. R. Andersen and B. G. Plosz, Diffusion and sorption of organic micropollutants in biofilms with varying thicknesses, Water Res., 2017, 123, 388–400 CrossRef CAS PubMed.
- T. A. Ternes, N. Herrmann, M. Bonerz, T. Knacker, H. Siegrist and A. Joss, A rapid method to measure the solid-water distribution coefficient (Kd) for pharmaceuticals and musk fragrances in sewage sludge, Water Res., 2004, 38(19), 4075–4084 CrossRef CAS PubMed.
- J. Tolls, Sorption of veterinary pharmaceuticals in soil: a review, Environ. Sci. Technol., 2001, 35(17), 3397–3406 CrossRef CAS PubMed.
- L. Berthod, G. Roberts and G. A. Mills, A solid-phase extraction approach for the identification of pharmaceutical–sludge adsorption mechanisms, J. Pharm. Anal., 2014, 4(2), 117–124 CrossRef.
- A. Pirovano, M. Huijbregts, A. Ragas, K. Veltman and A. J. Hendriks, Mechanistically-based QSARs to describe metabolic constants in mammals, Altern. Lab. Anim., 2014, 42, 59–69 CAS.
- C. Bellona and J. E. Drewes, The role of membrane surface charge and solute physico-chemical properties in the rejection of organic acids by NF membranes, J. Membr. Sci., 2005, 249(1–2), 227–234 CrossRef CAS.
- D. Camacho-Muñoz, J. Martín, J. L. Santos, I. Aparicio and E. Alonso, Occurrence of surfactants in wastewater: hourly and seasonal variations in urban and industrial wastewaters from Seville (Southern Spain), Sci. Total Environ., 2014, 468, 977–984 CrossRef PubMed.
- J. Yu, J. Hu, S. Tanaka and S. Fujii, Perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) in sewage treatment plants, Water Res., 2009, 43(9), 2399–2408 CrossRef CAS PubMed.
- N. Seetha, R. Bhargava and P. Kumar, Effect of organic shock loads on a two-stage activated sludge-biofilm reactor, Bioresour. Technol., 2010, 101(9), 3060–3066 CrossRef CAS PubMed.
- P. Guerra, M. Kim, A. Shah, M. Alaee and S. A. Smyth, Occurrence and fate of antibiotic, analgesic/anti-inflammatory, and antifungal compounds in five wastewater treatment processes, Sci. Total Environ., 2014, 473–474, 235–243 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c7em00493a |
| This journal is © The Royal Society of Chemistry 2018 |