Photolithography-compatible conformal electrodes for high-performance bottom-contact organic single-crystal transistors†
Abstract
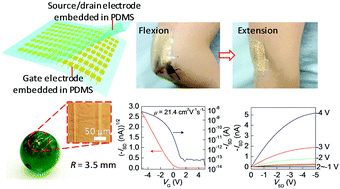
High-performance rubrene single-crystal field-effect transistors (SCFETs) with bottom-gate bottom-contact configuration were successfully fabricated on both planar and curved surfaces based on a photolithography-compatible conformal electrode. This electrode not only provides versatile precise patterns for device design, but also eliminates the device differences by the fabrication of multiple devices based on one single crystal, which is very favorable for studies of the intrinsic properties and integration of organic devices. The resulting rubrene SCFETs show excellent electrical properties with good device uniformity, zero hysteresis, a device yield as high as 92%, and a field-effect mobility of over 20 cm2 V−1 s−1 on different surfaces including a banknote, a pencil, and a 0.7 cm glass sphere. The high electrical performance in our bottom-contact devices can be attributed to the nondestructive interface contact and eliminated electrode steps. Such a soft coplanar electrode provides a preferred configuration for bottom-contact organic field-effect transistors (OFETs), facilitating the studies on the fundamental properties of organic transistors, and showing strong potential for the development of large-scale commercial organic transistor fabrication.


 Please wait while we load your content...
Please wait while we load your content...