0D/2D heterojunctions of molybdenum carbide-tungsten carbide quantum dots/N-doped graphene nanosheets as superior and durable electrocatalysts for hydrogen evolution reaction†
Abstract
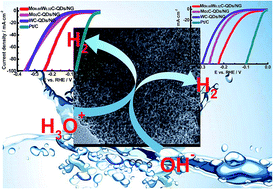
It is of great importance to exploit and design efficient and low-cost alternatives to platinum-based electrocatalysts for the hydrogen evolution reaction (HER). In this work, we report novel well-defined 0D/2D heterojunctions of uniform molybdenum carbide-tungsten carbide quantum dots ((Mo2C)x–(WC)1−x–QDs, ∼3–5 nm)/N-doped graphene (NG) nanosheets with a two-dimensional layered structure obtained via a nanocasting method using KIT-6/graphene (G) as a template. By controlling the molar ratio of the Mo and W precursors, (Mo2C)x–(WC)1−x–QDs (0 < x < 1)/NG nanohybrids with different Mo/W molar ratios can be obtained, which exhibit superior activity in the HER to individual Mo2C/NG and WC/NG nanohybrids. The superior activity in the HER may be attributed to redistribution of the valence electrons of Mo and W elements, nitrogen-coordinating sites, and highly dispersed Mo2C–WC nanocrystals, as well as strong coupling between Mo2C–WC nanocrystals and NG. Excitingly, the optimal electrocatalyst, namely, (Mo2C)0.34–(WC)0.32/NG, exhibited low overpotentials (100 and 93 mV) to achieve a cathodic current density of 10 mA cm−2, small Tafel slopes (53 and 53 mV dec−1), and high exchange current densities (0.419 and 0.804 mA cm−2) in acidic and alkaline media, respectively. More importantly, it also displayed excellent long-term durability for 25 h of stable catalytic current at different pH values. This work is expected to provide a feasible route for the fabrication of 0D/2D earth-abundant nanocomposites for the HER.


 Please wait while we load your content...
Please wait while we load your content...