Theoretical modulation of singlet/triplet chemiexcitation of chemiluminescent imidazopyrazinone dioxetanone via C8-substitution†
Abstract
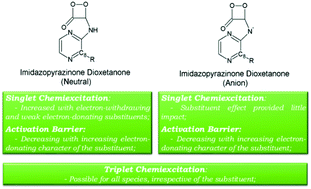
Coelenterazine, a member of the imidazopyrazinone class of chemiluminescent substrates, presents significant potential as a dynamic probe of reactive oxygen species in a biological environment, such as a superoxide anion, in which these species are important in cellular biology and pathology. The objective of the current study was to understand in what way the efficiency of singlet and triplet chemiexcitation could be modulated, towards a more efficient use of imidazopyrazinone-based compounds as dynamic chemiluminescent probes. To this end the thermolysis of imidazopyrazinone dioxetanone, substituted at the C8-position with electron-donating or electron-withdrawing groups, was characterized with a theoretical approach based on density functional theory. Substituents with different electron-donating/withdrawing characters have only a limited effect on the singlet chemiexcitation of anionic dioxetanone. For neutral dioxetanone, both electron-withdrawing and weak electron-donating substituents increase singlet chemiexcitation, to the contrary of strong electron-donating groups. During their thermolysis reaction, all molecules presented regions of degeneracy with triplet states, thereby indicating the possibility of triplet chemiexcitation.


 Please wait while we load your content...
Please wait while we load your content...