Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Chalcogenoether complexes of Nb(V) thio- and seleno-halides as single source precursors for low pressure chemical vapour deposition of NbS2 and NbSe2 thin films†
Yao-Pang
Chang
,
Andrew L.
Hector
 ,
William
Levason
,
William
Levason
 and
Gillian
Reid
and
Gillian
Reid
 *
*
Chemistry, University of Southampton, Southampton SO17 1BJ, UK. E-mail: G.Reid@soton.ac.uk
First published on 26th June 2017
Abstract
NbSCl3 was obtained via reaction of NbCl5 with S(SiMe3)2 in anhydrous CH2Cl2, whilst in MeCN solution the same reaction gives [NbSCl3(MeCN)2]. [NbSeCl3(MeCN)2] was obtained similarly from NbCl5 with Se(SiMe3)2. The chalcogenoether complexes, [NbSCl3(ER2)] (E = S: R = Me, nBu; E = Se: R = nBu), were obtained from reaction of NbCl5, ER2 and S(SiMe3)2 in CH2Cl2. The structure of the [Nb2S2Cl6(SMe2)2] reveals a Cl-bridged dimer with the SMe2 ligands disposed syn. The Cl bridges are highly asymmetric, with the long Nb–Cl bond trans Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S. The complexes, [NbSCl3(L–L)] (L–L = MeSCH2CH2SMe, MeS(CH2)3SMe, iPrSCH2CH2SiPr, MeSe(CH2)3SeMe and nBuS(CH2)3SnBu), were obtained from reaction of L–L with preformed [NbSCl3(MeCN)2]. The structures of the Me-substituted complexes reveal distorted octahedral monomers with the neutral ligands trans to S/Cl. Solution 1H and 77Se{1H} NMR data showed that the neutral ligands are partially dissociated and undergoing fast exchange at ambient temperatures in CH2Cl2 solution, consistent with weak Lewis acidity for NbSCl3. The complexes containing nBu-substituted ligands have been used as single source precursors for low pressure chemical vapour deposition (CVD) of 3R-NbS2 thin films. 2H-NbSe2 thin films were also obtained via low pressure CVD using [NbSe2Cl3(SenBu2)]. The thin films were characterised by grazing incidence and in-plane XRD, pole figure analysis, scanning electron microscopy and energy dispersive X-ray analysis.
S. The complexes, [NbSCl3(L–L)] (L–L = MeSCH2CH2SMe, MeS(CH2)3SMe, iPrSCH2CH2SiPr, MeSe(CH2)3SeMe and nBuS(CH2)3SnBu), were obtained from reaction of L–L with preformed [NbSCl3(MeCN)2]. The structures of the Me-substituted complexes reveal distorted octahedral monomers with the neutral ligands trans to S/Cl. Solution 1H and 77Se{1H} NMR data showed that the neutral ligands are partially dissociated and undergoing fast exchange at ambient temperatures in CH2Cl2 solution, consistent with weak Lewis acidity for NbSCl3. The complexes containing nBu-substituted ligands have been used as single source precursors for low pressure chemical vapour deposition (CVD) of 3R-NbS2 thin films. 2H-NbSe2 thin films were also obtained via low pressure CVD using [NbSe2Cl3(SenBu2)]. The thin films were characterised by grazing incidence and in-plane XRD, pole figure analysis, scanning electron microscopy and energy dispersive X-ray analysis.
Introduction
Layered early transition metal dichalcogenides ME2 (M = Nb, Ta, V, W etc., E = S, Se or Te), inorganic analogues of graphite or graphene, are highly stable, and their properties can be tuned by varying the metal and the chalcogen.1 Applications of these materials in areas such as optoelectronics, spintronics, sensors, electrocatalysis and magnetic materials are being actively developed.1–3 Production of the materials as thin films maximises the anisotropy of their magnetic or electronic properties and thus methods to deposit 2D layers are of great current interest. Chemical vapour deposition (CVD) is a low cost versatile method to deposit such films using either dual or single source precursors.4–6 Single source precursors potentially offer better control of deposit stoichiometry, cost-effective use of reagents, and in some cases the ability to selectively deposit the ME2 onto nano-patterned substrates. Reagents for CVD of NbS2 or NbSe2 are rare, but include niobium thiolate complexes for the former7 and a dual source approach using NbCl5-SetBu2 for the latter.6 We recently reported the deposition of thin films of NbS2 and NbSe2 as the 3R-polytype (R3mh) using [NbCl5(SnBu2)] and [NbCl5(SenBu2)], respectively, as single source low pressure chemical vapour deposition (LPCVD) precursors.8 The properties of the potential precursors are key to successful applications in this area, requiring appropriate volatility and clean deposition at accessible temperatures (a β-hydride decomposition route for the neutral ligand is often helpful). Our previous studies showed that Nb(III) dimers such as [Nb2Cl4(EnBu2)2(μ-Cl)2(μ-EnBu2)] did not vaporise under LPCVD conditions, probably due to their high molecular weights,9 whilst the Nb(IV) complexes such [NbCl4(SenBu2)2], [NbCl4{nBuSe(CH2)3SenBu}] and [NbCl4{iPrSCH2CH2SiPr}], lost the chalogenoether on heating in vacuo, leaving a residue of the (strongly polymerised) NbCl4.10Here we report on the synthesis and properties of chalcogenoether complexes of niobium chalcogenide trichlorides NbECl3 (E = S or Se) and attempts to use them as LPCVD reagents. Chalcogenohalides are known for many of the transition elements,11 although their coordination chemistry has been much less explored than those of the corresponding halides or oxide halides.12 Complexes of NbSX3 with Lewis bases include [NbSCl4]−,13 [NbSCl3(Ph3PS)],14 and [NbSX3(Ph3PO)2] (X = Cl, Br),15 [NbSCl3(SEt2)],15 and [NbSX3(MeCN)2],15,16 However, in other systems rearrangements occur to form compounds with dichalcogenide bridges, including the structurally characterised [Nb2Cl4(Se2)2(L)4] (L = SMe2 or tetrahydrothiophene (tht)) and [Nb2S3X4(tht)4].17,18
Results and discussion
Complex synthesis and properties
We obtained crude NbSCl3 as a dark green solid by reaction of NbCl5 with S(SiMe3)2 in CH2Cl2 in an ice-bath, and this solid was used to test the parent compound as an LPCVD reagent (see below). As pointed out by others,19 the colour of NbSCl3 is variously described as golden-yellow to green or black in the literature, and it was suggested that the darker coloured samples contain Nb2S3Cl4 impurities. The ν(Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S) vibration at 550 cm−1 agrees with the literature value.20
S) vibration at 550 cm−1 agrees with the literature value.20
The thio- and seleno-ether complexes of NbSCl3 were obtained in good yield either by reaction of NbCl5 with the ligand and S(SiMe3)2 in CH2Cl2 in an ice-bath, or by reaction of [NbSCl3(MeCN)2] with the ligand in CH2Cl2 at ambient temperatures. [NbSCl3(MeCN)2] was made by the literature method, involving reaction of NbCl5 with S(SiMe3)2 in MeCN solution,16 or by dissolving the crude NbSCl3 in MeCN, filtering to remove any insoluble material, and evaporating the filtrate to dryness. The products of the two methods are spectroscopically identical, and the data agree well with the literature.15,16
[NbSeCl3(MeCN)2] was obtained from NbCl5 and Se(SiMe3)2 in MeCN as a brown solid. The IR spectroscopic properties, ν(MeCN) = 2310, 2281, ν(Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se) = 397, ν(Nb–Cl) = 377, 344 cm−1, are as expected, although our assignment of Nb
Se) = 397, ν(Nb–Cl) = 377, 344 cm−1, are as expected, although our assignment of Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se and Nb–Cl vibrations is tentative as they occur at similar frequencies. The 93Nb NMR chemical shift, δ = 923, is at significantly higher frequency than in the thiochloride analogue, reflecting the effect of the selenium coordination.
Se and Nb–Cl vibrations is tentative as they occur at similar frequencies. The 93Nb NMR chemical shift, δ = 923, is at significantly higher frequency than in the thiochloride analogue, reflecting the effect of the selenium coordination.
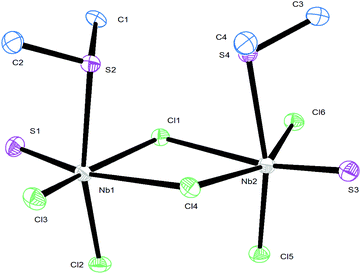
The reaction of NbCl5 with SMe2 and S(SiMe3)2 in CH2Cl2 cooled to ∼0 °C produced a yellow–green solid of stoichiometry [NbSCl3(SMe2)], whilst use of SnBu2 gave a dark oil [NbSCl3(SnBu2)]. The IR spectrum of [NbSCl3(SMe2)] shows a strong vibration at 530 cm−1, assigned as a terminal ν(Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), with ν(Nb–Cl) vibrations at 369, 356, 322 cm−1. In chlorocarbon solution it shows a single δ(Me) resonance in the 1H NMR spectrum, shifted to high frequency from SMe2, and, more interestingly, a 93Nb NMR resonance21 at δ = 651. The resonances of niobium thiochloride complexes are found to high frequency of those of NbCl5.22,23 (Note that the chemical shifts in ref. 22 are reported using the old high frequency negative convention.) The spectroscopic data on [NbECl3(SnBu2)] were similar, suggesting an analogous structure. The X-ray structure of [NbSCl3(SMe2)] showed it to be a dimer, [Nb2S2Cl4(μ-Cl)2(SMe2)2] (Fig. 1). The bond lengths are unexceptional, with the exception that those associated with the bridging Cl ligands lying trans to Nb–S are highly asymmetric. Notably the two SMe2 ligands are syn-disposed, rather than the much more common anti arrangement found in many centrosymmetric d-block dimers.
S), with ν(Nb–Cl) vibrations at 369, 356, 322 cm−1. In chlorocarbon solution it shows a single δ(Me) resonance in the 1H NMR spectrum, shifted to high frequency from SMe2, and, more interestingly, a 93Nb NMR resonance21 at δ = 651. The resonances of niobium thiochloride complexes are found to high frequency of those of NbCl5.22,23 (Note that the chemical shifts in ref. 22 are reported using the old high frequency negative convention.) The spectroscopic data on [NbECl3(SnBu2)] were similar, suggesting an analogous structure. The X-ray structure of [NbSCl3(SMe2)] showed it to be a dimer, [Nb2S2Cl4(μ-Cl)2(SMe2)2] (Fig. 1). The bond lengths are unexceptional, with the exception that those associated with the bridging Cl ligands lying trans to Nb–S are highly asymmetric. Notably the two SMe2 ligands are syn-disposed, rather than the much more common anti arrangement found in many centrosymmetric d-block dimers.
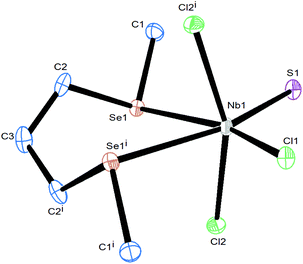
The MeCN ligands in [NbSCl3(MeCN)2] were cleanly substituted on reaction with an excess (three equivalents) of MeSCH2CH2SMe, MeS(CH2)3SMe, iPrSCH2CH2SiPr or MeSe(CH2)3SeMe in CH2Cl2 solution at ambient temperatures, to give green [NbSCl3(L–L)] (L–L = MeSCH2CH2SMe, MeS(CH2)3SMe, iPrSCH2CH2SiPr) or brown [NbSCl3{MeSe(CH2)3SeMe}] powdered solids. Using nBuS(CH2)3SnBu produced [NbSCl3{nBuS(CH2)3SnBu}] as a green oil. The X-ray crystal structure of [NbSCl3{MeSe(CH2)3SeMe}] showed a distorted octahedral geometry, with the in plane terminal S/Cl disordered. The disorder was successfully modelled using split sites for these two atoms and refined at 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 occupancies; only one form is shown in Fig. 2. The key bond lengths are Nb1–Cl1 = 2.308(7), Nb1–S1 = 2.197(8) Å, whilst the axial Nb1–Cl2 = 2.3686(6) Å, i.e. significantly longer.
50 occupancies; only one form is shown in Fig. 2. The key bond lengths are Nb1–Cl1 = 2.308(7), Nb1–S1 = 2.197(8) Å, whilst the axial Nb1–Cl2 = 2.3686(6) Å, i.e. significantly longer.
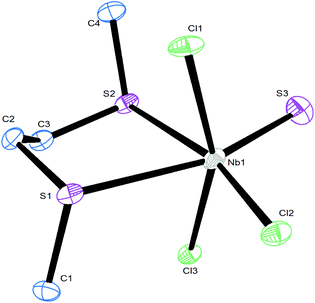
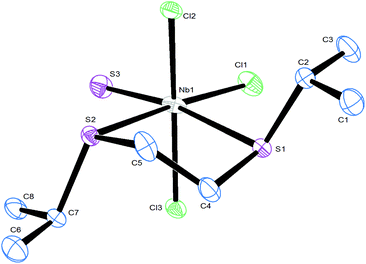
The structures of two thioether complexes, [NbSCl3(MeSCH2CH2SMe)] and [NbSCl3(iPrSCH2CH2SiPr)], were also determined and are shown in Fig. 3 and 4. The X-ray diffraction data were good quality and careful examination did not provide clear evidence for S/Cl disorder in these cases and the d(Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S) = 2.10 Å and d(Nb–Cl) = 2.24 Å are nearly identical in the two structures. Notably, the d(Nb–S) distances trans to the Nb
S) = 2.10 Å and d(Nb–Cl) = 2.24 Å are nearly identical in the two structures. Notably, the d(Nb–S) distances trans to the Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S and trans to Nb–Cl are different, consistent with a trans influence of Nb
S and trans to Nb–Cl are different, consistent with a trans influence of Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S > Nb–Cl. However, as pointed out by others,14,17,18 it is not possible to rule out some disorder in the thiochloride systems due to the very similar scattering power of S and Cl atoms. We note in passing that all three structures contain the dichalcogenoether in the DL form.
S > Nb–Cl. However, as pointed out by others,14,17,18 it is not possible to rule out some disorder in the thiochloride systems due to the very similar scattering power of S and Cl atoms. We note in passing that all three structures contain the dichalcogenoether in the DL form.
The IR spectra of the five complexes exhibit ν(Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S) ∼ 525 cm−1 and two ν(Nb–Cl) stretches in the range 350–320 cm−1, sometimes with a shoulder on the higher energy vibration, indicating the same molecular geometry. As expected, they are diamagnetic, confirming the presence of d0 Nb(V). The 93Nb NMR spectra each exhibit a broad resonance in the region δ = +500 to +550, significantly to high frequency of NbOCl3 or NbCl5–thioether complexes.10,23 The 1H NMR spectrum of [NbSCl3(MeSCH2CH2SMe)] in CD2Cl2 at 295 K shows two very broad resonances assignable to δ(MeS) and two δ(CH2) resonances, which upon cooling the solution resolve into resonances due to free dithioether24 and coordinated dithioether, and at 183 K some further splitting is evident, probably due to slowing of pyramidal inversion at the sulfur centres. The data are consistent with some dissociation of the dithioether in solution, and fast exchange between coordinated and ‘free’ dithioether at ambient temperature. This is further evidence for the weaker Lewis acidity of NbSCl3 compared to NbOCl3 or NbCl5. The 1H NMR spectra of the other four complexes also have broad resonances at ambient temperatures, and show similar changes on cooling the samples. The complex [NbSCl3(MeSeCH2CH2CH2SeMe)] does not exhibit a 77Se{1H} NMR resonance at 295 K, but on cooling to 223 K, resonances due to the ‘free’ diselenoether (δ = 68),25 and coordinated diselenoether (δ = 163, 70) appear. The singlet at 70 ppm seems anomalous, but is reproducible and may reflect the ‘free’ Se donor of a κ1-coordinated diselenoether, although it is possible this arises due to the Se donor atom trans to the terminal S.
S) ∼ 525 cm−1 and two ν(Nb–Cl) stretches in the range 350–320 cm−1, sometimes with a shoulder on the higher energy vibration, indicating the same molecular geometry. As expected, they are diamagnetic, confirming the presence of d0 Nb(V). The 93Nb NMR spectra each exhibit a broad resonance in the region δ = +500 to +550, significantly to high frequency of NbOCl3 or NbCl5–thioether complexes.10,23 The 1H NMR spectrum of [NbSCl3(MeSCH2CH2SMe)] in CD2Cl2 at 295 K shows two very broad resonances assignable to δ(MeS) and two δ(CH2) resonances, which upon cooling the solution resolve into resonances due to free dithioether24 and coordinated dithioether, and at 183 K some further splitting is evident, probably due to slowing of pyramidal inversion at the sulfur centres. The data are consistent with some dissociation of the dithioether in solution, and fast exchange between coordinated and ‘free’ dithioether at ambient temperature. This is further evidence for the weaker Lewis acidity of NbSCl3 compared to NbOCl3 or NbCl5. The 1H NMR spectra of the other four complexes also have broad resonances at ambient temperatures, and show similar changes on cooling the samples. The complex [NbSCl3(MeSeCH2CH2CH2SeMe)] does not exhibit a 77Se{1H} NMR resonance at 295 K, but on cooling to 223 K, resonances due to the ‘free’ diselenoether (δ = 68),25 and coordinated diselenoether (δ = 163, 70) appear. The singlet at 70 ppm seems anomalous, but is reproducible and may reflect the ‘free’ Se donor of a κ1-coordinated diselenoether, although it is possible this arises due to the Se donor atom trans to the terminal S.
Attempts to prepare dithio- or diseleno-ether complexes from [NbSeCl3(MeCN)2], even using excess chalcogenoethers or long reaction times, were unsuccessful. However, the reaction of NbCl5 with SenBu2 in CH2Cl2, followed by treatment with (Me3Si)2Se, gave a black solid which, on the basis of microanalysis and IR and 1H NMR spectroscopic analysis, was tentatively identified as [NbSe2Cl3(SenBu2)]. The structure of this material is unknown, but it may be dimeric like [Nb2Cl4(Se2)2(SMe2)2] and contain [Se–Se]2− groups.17
Application for LPCVD of NbE2 thin films
Although the gas phase structure of [NbSCl3] has been determined,19 indicating some volatility, attempts to use this as a precursor for deposition of NbS2 in the temperature range 400–750 °C were unsuccessful. However, the complexes, [NbSCl3(SnBu2)], [NbSCl3{nBuS(CH2)3SnBu}] and [NbSe2Cl3(SenBu2)], were likely candidates for use as single source CVD precursors for the low pressure CVD of NbS2 and NbSe2 thin films, owing to their volatility and the presence of readily eliminated nBu groups. Thermogravimetric analyses were undertaken for these complexes (ESI Fig. S4–S6†), and while the mass loss observed does not correspond with the residue being simply NbS2 or NbSe2, this is unsurprising given the different experimental conditions used for TGA (progressive heating under a flowing argon atmosphere) compared to the vapour transport process involved in the low pressure CVD experiments. However, the TGA experiments do indicate the temperature at which mass loss begins, providing an indication of the low temperature threshold. Low pressure CVD experiments were therefore performed at a pressure of ca. 0.05 mmHg using a range of temperatures around 600–750 °C. No significant deposition was observed at lower temperatures.Black reflective films were obtained from [NbSCl3(SnBu2)] at 700 °C, and grazing incidence XRD showed patterns consistent with NbS2 in space group R3mh (3R-type NbS2) (Fig. 5). These films appear to be air and moisture stable, and lattice parameters determined by Rietveld refinement of the grazing incidence XRD pattern are: a = 3.317(6) and c = 17.79(4) Å (Rwp = 6.61%, Rp = 4.46%). These are close to literature values for bulk NbS2 (a = 3.3303(3), c = 17.918(2) Å).26 The grazing incident and in-plane XRD patterns shows considerable variations in the intensities of reflections relative to the bulk pattern. The 003 reflection is the strongest in the grazing incidence XRD pattern, and 101, 012 and 110 reflections are highest in the in-plane XRD pattern, suggesting <00l> preferred orientation.
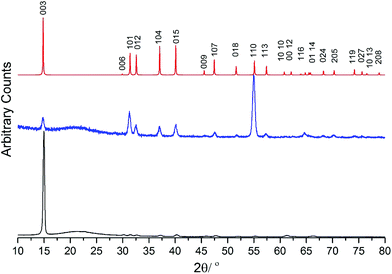 | ||
| Fig. 5 In plane XRD (blue), grazing incidence (incidence angle = 1°) XRD (black) from the NbS2 thin film deposited by LPCVD using [NbSCl3(SnBu2)] at 700 °C; simulated XRD pattern from bulk NbS2 (red).26 The broad feature at 2θ ∼22° is from the SiO2 substrate. | ||
Pole figure measurements were undertaken on a NbS2 film obtained from the [NbSCl3(SnBu2)] precursor to establish the film texture. Using 2θ = 14.97°, corresponding to the 0 0 3 reflection, a single, sharp peak (FWHM ∼ 5°) was observed at the centre of the figure with α = 90° (Fig. 6a). The figure taken with 2θ = 31.47°, corresponding to the 1 0 1, exhibits a ring with α = 9° (Fig. 6b). These results confirmed the <00l> preferred orientation in which the ab planes of the crystallites are parallel with the substrate.
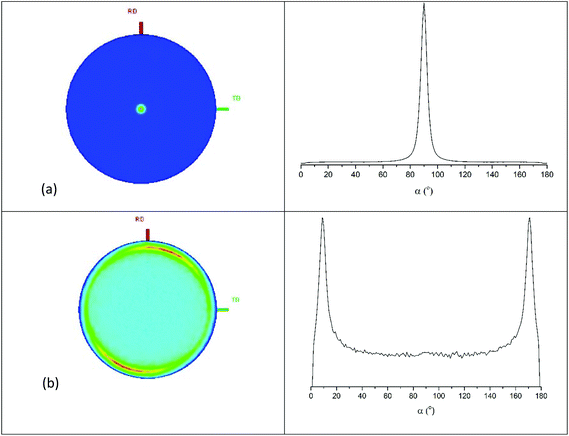 | ||
| Fig. 6 Pole figures with cut line integral graphs for the 0 0 3 (2θ = 14.97°) (a) and 1 0 1 (2θ = 31.47°) (b) reflections of a film of NbS2 deposited on a SiO2 substrate. | ||
Scanning electron microscopy (SEM) images reveal that the NbS2 films have a uniform morphology formed of microcrystalline platelets mainly lying flat on the substrate (Fig. 7), consistent with the orientation inferred from the XRD data. EDX data measured at an accelerating voltage of 15 keV show significant Si and O peaks in addition to peaks due to Nb and S, indicating that the films are thin. EDX data also shows there is no evidence for any residual Cl in the films (Cl Kα = 2.621 keV). Accurate quantification of the Nb![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S ratio by EDX is difficult due to the Nb Lα and S Kα peaks overlapping.
S ratio by EDX is difficult due to the Nb Lα and S Kα peaks overlapping.
Films obtained from low pressure CVD using [NbSCl3{nBuS(CH2)3SnBu}] at 700 °C also present diffraction patterns consistent with R3mh (3R-type NbS2) (Fig. S1†), with lattice parameters a = 3.29(2) and c = 17.8(2) Å (Rwp = 5.54%, Rp = 3.47%). SEM images of these films are presented in the ESI (Fig. S2†).
Low pressure CVD experiments were also performed using [NbSe2Cl3(SenBu2)] at temperatures between 600 and 700 °C (0.05 mmHg). Films obtained at 650 °C present diffraction patterns consistent with P63/mmc (2H-type NbSe2) (Fig. 8). Previously we obtained 3R-NbSe2 from [NbCl5(SenBu2)].8 but Parkin and co-workers have reported 2H-NbSe2 by dual source APCVD from NbCl5 and SetBu2.6 The 002 101 and 110 reflections are the three most intense in both grazing incidence and in-plane XRD patterns, indicating that preferred orientation is not significant in this case. However, using a reduced amount of precursor produces thinner films with evidence of preferred orientation (Fig. S3†). Lattice parameters determined by Rietveld refinement of the grazing incidence XRD pattern are: a = 3.434(7) and c = 12.53(3) Å (Rwp = 2.48%, Rp = 1.87%), comparing to the literature values for bulk NbSe2 of a = 3.4446(2), c = 12.5444(7) Å.27 The NbSe2 films appear to be air and moisture stable. The precursor was also tested in LPCVD at 600 °C however, there is no deposition observed. Scanning electron microscopy (SEM) of the same NbSe2 film shows a polycrystalline film formed of hexagonal platelets mostly oriented with the c axis parallel to the substrate (Fig. 9), although the absence of significant preferred orientation from the XRD data suggests it is likely that there are different crystal orientations within the film. Energy dispersive X-ray spectroscopy (EDX) gives the ratio of Nb![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Se in the films of 35.8%
Se in the films of 35.8%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64.2%, with no detectable Cl contamination.
64.2%, with no detectable Cl contamination.
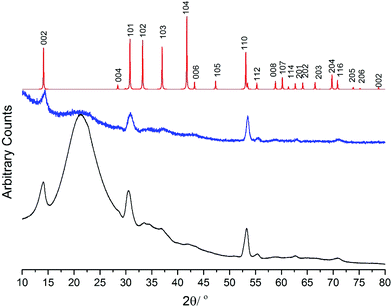 | ||
| Fig. 8 In plane XRD (blue), grazing incidence (incidence angle = 1°) XRD (black) from the NbSe2 thin film deposited by LPCVD using [NbSe2Cl3(SenBu2)] at 650 °C; simulated XRD pattern from bulk NbSe2 (red).27 The broad feature at 2θ ∼22° is from the SiO2 substrate. | ||
Conclusions
A series of thioether and selenoether complexes derived from NbSCl3 is reported. NbSCl3 has been shown to be a weaker Lewis acid than NbCl5. Selected complexes containing nBu substituents have been used as single source precursors to produce 3R-NbS2 thin films via low pressure CVD, with preferred <00l> orientation. In contrast, NbSeCl3 does not form similar complexes, however treatment of [NbCl5(SenBu2)] with Se(SiMe3)2 forms the complex [NbSe2Cl3(SenBu2)] which has been used as a single source precursor for low pressure CVD of 2H-NbSe2 thin films.Experimental
Syntheses were performed by using standard Schlenk and glove-box techniques under a dry N2 atmosphere. NbCl5 and S(SiMe3)2 were obtained from Sigma-Aldrich and Se(SiMe3)2 from Fluorochem and stored in the glovebox in ampoules under N2. Solvents were dried by distillation from CaH2 (CH2Cl2, MeCN) or Na/benzophenone ketyl (n-hexane). Dichalcogenoethers, RE(CH2)nER (E = S, Se; n = 2, 3; R = Me, iPr, nBu), were prepared via literature methods.24,25Physical measurements
Infrared spectra were recorded on a PerkinElmer Spectrum 100 spectrometer in the range 4000–200 cm−1, and samples prepared as Nujol mulls between two CsI plates. 1H NMR spectra were recorded using a Bruker AV II 400 spectrometer and referenced to the residual protio-resonance of the solvent. Multinuclear (77Se, 93Nb) NMR spectra were recorded from CD2Cl2, CDCl3 or CD3CN solutions using a Bruker AV II 400 spectrometer and referenced to neat external SeMe2 and [Et4N][NbCl6] in MeCN (δ = 0), respectively. Microanalyses on new compounds were undertaken by London Metropolitan University. Thermogravimetric analysis (TGA) data for [NbSCl3(SnBu2)] (ESI Fig. S4†), [NbSCl5(nBuSCH2CH2CH2SnBu)] (ESI Fig. S5†) and [NbSe2Cl3(SenBu2)] (ESI Fig. 6†) were collected via a Netzsch TG209 F1 Libra analyser under a flow of argon at 65 mL min−1, contained within a dry, nitrogen purged glovebox. The temperature was increased at a rate of 10 °C min−1. Samples were loaded in Al sample pans.X-ray experimental
Data collections used a Rigaku AFC12 goniometer equipped with an enhanced sensitivity (HG) Saturn724+ detector mounted at the window of an FR-E+ SuperBright molybdenum (λ = 0.71073) rotating anode generator with VHF Varimax optics (70 micron focus) with the crystal held at 100 K (N2 cryostream). Crystallographic parameters are in the ESI (Table S1†). Structure solution and refinement were performed using SHELX(S/L)97, SHELX-2014/7 and were mostly straightforward, except for the Cl and terminal S atoms of the [NbSCl3{MeSe(CH2)3SeMe}] which were disordered and therefore refined with split occupancies giving a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio.28,29 H atoms were added and refined with a riding model.
50 ratio.28,29 H atoms were added and refined with a riding model.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S).
S).
[NbSCl
3
(MeCN)
2
]: Method 1: The complex was prepared by a modified literature method.16 NbCl5 (405 mg, 1.5 mmol) was dissolved in MeCN (30 mL) and cooled in an ice bath. A solution of (Me3Si)2S (260 mg, 1.5 mmol) in MeCN (20 mL) was added. The reaction mixture was then removed from the ice bath and the colour changed from yellow to green over a few minutes. The solution was stirred for 1 h giving a green solution. The solvent was then removed in vacuo leaving a yellow–green solid. Yield: 337 mg, 72%. Required for C4H6N2Cl3NbS (313.33): C, 15.33; H, 1.93; N, 8.94. Found: C, 15.26; H, 1.96; N, 8.82%. IR (Nujol, cm−1): 2316, 2287s (MeCN), 530s (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 355sh, 343s, 319s (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 1.97 (s, MeCN). 93Nb NMR (CD3CN, 295 K): δ = 414.
S), 355sh, 343s, 319s (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 1.97 (s, MeCN). 93Nb NMR (CD3CN, 295 K): δ = 414.
Method 2: NbSCl3 (83 mg, 0.35 mmol) was dissolved in MeCN (20 mL) with stirred for 30 min and giving a dark green solution. The solution was filtered and the filtrate taken to dryness in vacuo. Green powder. Yield: 100 mg, 90%. The product was spectroscopically identical to that of [NbSCl3(MeCN)2] produced by Method 1.
[NbSCl
3
(SMe
2
)]: NbCl5 (135 mg, 0.5 mmol) was suspended in CH2Cl2 (10 mL). SMe2 (1 mL) was added with stirring for 30 min, giving a dark brown solution. The solution was then cooled in an ice bath (0 °C) and a solution of S(SiMe3)2 (90 mg, 0.5 mmol) in CH2Cl2 (ca. 1 mL) was added slowly with stirring for 30 min. The solution was removed from ice bath and stirred for another 5 min and then the volatiles were removed in vacuo, leaving a light yellow green solid. Yield: 55 mg, 38%. Required for C2H6Cl3NbS2 (293.46): C, 9.19; H, 2.06. Found: C, 9.29; H, 2.37%. IR (Nujol, cm−1): 530 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 369, 356, 322 (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 2.32 (s, SMe2). 93Nb NMR (CD2Cl2, 295 K): δ = 651. Yellow–green crystals grew by allowing CH2Cl2 solution to evaporate slowly under a nitrogen atmosphere.
S), 369, 356, 322 (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 2.32 (s, SMe2). 93Nb NMR (CD2Cl2, 295 K): δ = 651. Yellow–green crystals grew by allowing CH2Cl2 solution to evaporate slowly under a nitrogen atmosphere.
[NbSCl
3
(S
n
Bu
2
)]: NbCl5 (270 mg, 1.0 mmol) was suspended in CH2Cl2 (10 mL) and a solution of SnBu2 (147 mg, 1.0 mmol) in CH2Cl2 (3 mL) was added with stirring. After 15 min an orange solution formed. A solution of S(SiMe3)2 (179 mg, 1.0 mmol) and CH2Cl2 (3 mL) was added with stirring. The colour changed to dark green after stirring for 30 min. The solution was taken to dryness in vacuo, leaving a sticky oil which was washed with n-hexane (5 mL), before drying in vacuo. Thick dark oil. Yield: 331 mg, 88%. Required for C8H18Cl3NbS2 (377.62): C, 25.44; H, 4.80. Found: C, 25.58; H, 4.88%. IR (Nujol, cm−1): 554 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 387, 374, 359sh (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 0.97 (t, [6H], Me), 1.48 (m, [4H], CH2Me), 1.74 (m, [4H], SCH2CH2CH2), 2.94 (t, [4H], SCH2). 93Nb NMR (CD2Cl2, 295 K): δ = 654.
S), 387, 374, 359sh (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 0.97 (t, [6H], Me), 1.48 (m, [4H], CH2Me), 1.74 (m, [4H], SCH2CH2CH2), 2.94 (t, [4H], SCH2). 93Nb NMR (CD2Cl2, 295 K): δ = 654.
[NbSCl
3
(Se
n
Bu
2
)]: NbCl5 (270 mg, 1.0 mmol) was suspended in CH2Cl2 (10 mL) and a solution of SenBu2 (193 mg, 1.0 mmol) in CH2Cl2 (3 mL) was added with stirring, giving a red solution, which was stirred for 1 h. S(SiMe3)2 (0.21 mL, 1.0 mmol) was then added. The solution turned dark green, and after stirring for 30 min the solvent was removed in vacuo to leave a sticky oil. After washing with hexane (5 mL), the oil was dried in vacuo. Sticky black oil. Yield: 200 mg, 47%. Required for C8H18Cl3NbSSe (424.52): C, 22.63; H, 4.27. Found: C, 22.52; H, 4.29%. IR (Nujol, cm−1): 530 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 380, 366, 355sh (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 0.93 (t, [6H], Me), 1.42 (m, [4H], CH2Me), 1.66 (m, [4H], SeCH2CH2), 2.61 (br, [4H], SeCH2). 77Se (CD2Cl2, 295 K): no resonance. 93Nb NMR (CD2Cl2, 295 K): δ = 694.
S), 380, 366, 355sh (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 0.93 (t, [6H], Me), 1.42 (m, [4H], CH2Me), 1.66 (m, [4H], SeCH2CH2), 2.61 (br, [4H], SeCH2). 77Se (CD2Cl2, 295 K): no resonance. 93Nb NMR (CD2Cl2, 295 K): δ = 694.
[NbSCl
3
(MeSCH
2
CH
2
SMe)]: [NbSCl3(MeCN)2] (94 mg, 0.3 mmol) was dissolved in CH2Cl2 (10 mL) at room temperature. A solution of MeSCH2CH2SMe (110 mg, 0.9 mmol) and CH2Cl2 (1 mL) was added and the solution stirred for 30 min. Some green-yellow powder formed in the solution. After filtering, the green solution was evaporated to dryness in vacuo. n-Hexane (10 mL) was added to wash the solid which was separated and dried in vacuo. Yield: 67 mg, 63%. Required for C4H10Cl3NbS3 (353.58): C, 13.59; H, 2.85. Found: C, 13.64; H, 2.93%. IR (Nujol, cm−1): 526 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 361sh, 349, 319 (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 2.22 (br, [3H], SMetrans Cl), 2.78 (br, [3H], SMetrans S) 3.04(s), 3.26(s) ([4H], CH2); (183 K): 2.06 (s, [6H], Me), 2.63 (s, [4H], CH2) (both ‘free’ dithoether), 2.12 (s, [3H]), 2.18 (s, [3H]), 2.76, 2.94 (CH2) (coord. dithioether) (see text). 93Nb NMR (CD2Cl2, 295 K): δ = 507. Green crystals grew by allowing a CH2Cl2 solution to evaporate under a dinitrogen atmosphere.
S), 361sh, 349, 319 (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 2.22 (br, [3H], SMetrans Cl), 2.78 (br, [3H], SMetrans S) 3.04(s), 3.26(s) ([4H], CH2); (183 K): 2.06 (s, [6H], Me), 2.63 (s, [4H], CH2) (both ‘free’ dithoether), 2.12 (s, [3H]), 2.18 (s, [3H]), 2.76, 2.94 (CH2) (coord. dithioether) (see text). 93Nb NMR (CD2Cl2, 295 K): δ = 507. Green crystals grew by allowing a CH2Cl2 solution to evaporate under a dinitrogen atmosphere.
[NbSCl
3
(
i
PrSCH
2
CH
2
S
i
Pr)]: Was made similarly and isolated as a green powder. Yield: 76%. Required for C8H18Cl3NbS3 (409.69): C, 23.45; H, 4.43. Found: C, 23.27; H, 4.41%. IR (Nujol, cm−1): 527 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 365 (sh), 348, 318 (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 1.32 (br s, [6H], SMe), 1.59 (br s, [6H], SMe), 3.01–3.28 (br, [4H], SCH2), 3.46 (br, [2H], SCH). 93Nb NMR (CD2Cl2, 295 K): δ = 523. Green crystals grew by allowing a CH2Cl2 solution to evaporate slowly under a dinitrogen atmosphere.
S), 365 (sh), 348, 318 (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 1.32 (br s, [6H], SMe), 1.59 (br s, [6H], SMe), 3.01–3.28 (br, [4H], SCH2), 3.46 (br, [2H], SCH). 93Nb NMR (CD2Cl2, 295 K): δ = 523. Green crystals grew by allowing a CH2Cl2 solution to evaporate slowly under a dinitrogen atmosphere.
[NbSCl
3
{MeS(CH
2
)
3
SMe}]: Was made similarly and isolated as a green solid. Yield: 68% Required for C5H12Cl3NbS3 (367.61): C, 16.34; H, 3.29. Found: C, 16.48; H, 3.21%. IR (Nujol, cm−1): 524 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 369 sh, 345, 323 (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 2.14 (br, [2H], SCH2CH2), 2.45 (vbr, [6H], SMe), 2.96 (vbr, [4H], SCH2). 93Nb NMR (CD2Cl2, 295 K): δ = 530.
S), 369 sh, 345, 323 (Nb–Cl). 1H NMR (CDCl3, 295 K): δ = 2.14 (br, [2H], SCH2CH2), 2.45 (vbr, [6H], SMe), 2.96 (vbr, [4H], SCH2). 93Nb NMR (CD2Cl2, 295 K): δ = 530.
[NbSCl
3
{
n
BuS(CH
2
)
3
S
n
Bu}]: Was made similarly and isolated as a dark green oil after washing with n-hexane, decanting off the washings and drying in vacuo. Yield: 76%. Required for C11H24Cl3NbS3 (451.77): C, 29.24; H, 5.35. Found: C, 29.37; H, 5.45%. IR (Nujol, cm−1): 529 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 349, 322 (Nb–Cl). 93Nb NMR (CD2Cl2, 295 K): δ = 534.
S), 349, 322 (Nb–Cl). 93Nb NMR (CD2Cl2, 295 K): δ = 534.
[NbSCl
3
{MeSe(CH
2
)
3
SeMe}]: Was made similarly and isolated as a yellow brown powder. Yield: 58%. Required for C5H12Cl3NbSSe2 (461.40): C, 13.02; H, 2.62. Found: C, 13.17; H, 2.74%. IR (Nujol, cm−1): 521 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), 342, 320 (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 2.25 (br, [8H], SeMe and CH2CH2Se), 2.92 (br, [4H], CH2Se). 77Se{1H} NMR (CD2Cl2, 295 K): no resonance; (223 K): δ = 163, 70, 68. 93Nb NMR (CD2Cl2, 295 K): δ = 547. Yellow crystals grew by allowing a CH2Cl2 solution to evaporate under a dinitrogen atmosphere.
S), 342, 320 (Nb–Cl). 1H NMR (CD2Cl2, 295 K): δ = 2.25 (br, [8H], SeMe and CH2CH2Se), 2.92 (br, [4H], CH2Se). 77Se{1H} NMR (CD2Cl2, 295 K): no resonance; (223 K): δ = 163, 70, 68. 93Nb NMR (CD2Cl2, 295 K): δ = 547. Yellow crystals grew by allowing a CH2Cl2 solution to evaporate under a dinitrogen atmosphere.
[NbSeCl
3
(MeCN)
2
]: NbCl5 (135 mg, 0.5 mmol) was dissolved in MeCN (10 mL). A solution of Se(SiMe3)2 (113 mg, 0.5 mmol) in MeCN (5 mL) was then added and the reaction mixture was stirred for 30 min. The colour changed to dark brown. The solvent was removed in vacuo leaving a brown solid. Yield: 150 mg, 83%. Required for C4H6Cl3N2NbSe (360.33): C, 13.33; H, 1.68; N, 7.77. Found: C, 13.25; H, 1.65; N, 7.57%. IR (Nujol, cm−1): 2310, 2281 (MeCN), 397 (Nb![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se?), 377, 344 (Nb–Cl). 93Nb NMR (CD2Cl2, 295 K): δ = 923.
Se?), 377, 344 (Nb–Cl). 93Nb NMR (CD2Cl2, 295 K): δ = 923.
[NbSe 2 Cl 3 (Se n Bu 2 )]: NbCl5 (235 mg, 0.88 mmol) was suspended in CH2Cl2 (20 mL). A solution of nBu2Se (177 mg, 0.88 mmol) and CH2Cl2 (ca. 1.5 mL) was added with stirring for 1 hour. The reaction mixture changed colour to dark red and all the NbCl5 dissolved. A solution of Se(SiMe3)2 (0.22 mL, 0.88 mmol) and CH2Cl2 (ca. 1.7 mL) was added, causing a colour change from dark red to black. The solution was stirred for 15 min before it was taken to dryness in vacuo. The resulting black solid was washed with n-hexane (15 mL) and then dried in vacuo. Yield: 298 mg, 61%. Required for C8H18Cl3NbSe3 (550.37): C, 17.46; H, 3.3. Found: C, 17.59; H, 3.38. IR (Nujol, cm−1): 344, 319, 272 (Nb–Cl). 1H NMR (CDCl3, 295 K): δ 0.94 (t, [6H], Me), 1.44 (m, [4H], CH2Me), 1.70 (m, [4H], SeCH2CH2CH2), 2.71 (t, [4H], SeCH2).
LPCVD from [NbSCl3(SnBu2)], [NbSCl3{nBuS(CH2)3SnBu}] and [NbSe2Cl3(SenBu2)] resulted in deposition of reflective black films on tiles positioned in the hotter region of the furnace (i.e. NbS2 actual deposition temperature ∼670 °C; NbSe2 actual deposition temperature ∼625 °C – measured by profiling the furnace).
Scanning electron microscopy (SEM) was performed on samples of NbSe2 using a Philips XL30 ESEM and with an acceleration voltage of 10 kV or 15 kV, whilst SEM measurements on NbS2 films used a JEOL JSM6500 and an accelerating voltage of 10 kV.
Energy dispersive X-ray (EDX) data on NbSe2 samples were obtained at accelerating voltage of 10 and 15 kV with a Thermofisher Ultradry NSS 3 (XL30) detector or at an accelerating voltage of 15 kV with an Oxford INCA x-act X-ray detector (JSM6500) for NbS2 samples.
Acknowledgements
We thank the EPSRC for funding the SmartLab diffractometer (via EP/K00509X/1 and EP/K009877/1) and the University of Southampton for a VC Scholarship (to Y.-P. C).References
- M. Chhowalla, Z. Liu and H. Zhang, (Eds. - themed issue on metal dichalcogenides) Chem. Soc. Rev., 2015, 44, 2584.
- M. Chhowalla, H. S. Shin, G. Eda, L.-J. Li, K. P. Loh and H. Zhang, Nat. Chem., 2013, 5, 263 CrossRef CAS PubMed
; J. Liu and X.-W. Liu, Adv. Mater., 2012, 24, 4097 CrossRef PubMed
.
- Q. Xiang, J. Yu and M. Jaroniec, J. Am. Chem. Soc., 2012, 134, 6575 CrossRef CAS PubMed
; J. Pu, Y. Yomogida, K.-K. Liu, L.-J. Li, Y. Iwasa and T. Takenobu, Nano Lett., 2012, 12, 4013 CrossRef PubMed
; K. Lee, R. Gatensby, N. McEvoy, T. Hallam and G. S. Duesberg, Adv. Mater., 2013, 25, 6699 CrossRef PubMed
; Y. Ma, Y. Dsi, M. Guo, C. Niu, Y. Zhu and B. Huang, ACS Nano, 2012, 6, 1695 CrossRef PubMed
; K. Xu, P. Chen, X. Li, C. Wu, Y. Guo, J. Zhao, X. Wu and Y. Xie, Angew. Chem., Int. Ed., 2013, 52, 10477 CrossRef PubMed
; H. Li, G. Lu, Y. Wang, Z. Yin, C. Cong, Q. He, L. Wang, F. Ding, T. Yu and H. Zhang, Small, 2013, 9, 1974 CrossRef PubMed
; Z. Yan, C. Jiang, T. R. Pope, C. F. Tsang, J. L. Stickney, P. Goli, J. Renteria, T. T. Salguero and A. A. Balandin, J. Appl. Phys., 2013, 114, 20430 Search PubMed
.
-
A. C. Jones and M. L. Hitchman, in Chemical Vapour Deposition: Precursors, Processes and Applications, ed. A. C. Jones and M. L. Hitchman, The Royal Society of Chemistry, 2009 Search PubMed
.
- P. J. McKarns, T. S. Lewkebandara, G. P. A. Yap, L. M. Liable-Sands, A. L. Rheingold and C. H. Winter, Inorg. Chem., 1998, 37, 418 CrossRef CAS PubMed
; S. D. Reid, A. L. Hector, W. Levason, G. Reid, B. J. Waller and M. Webster, Dalton Trans., 2007, 4769 RSC
; S. L. Benjamin, C. H. de Groot, C. Gurnani, A. L. Hector, R. Huang, K. Ignatyev, W. Levason, S. J. Pearce, F. Thomas and G. Reid, Chem. Mater., 2013, 25, 4719 CrossRef PubMed
; N. D. Boscher, C. J. Carmalt and I. P. Parkin, Chem. Vap. Deposition, 2006, 12, 54 CrossRef
.
- N. D. Boscher, C. J. Carmalt and I. P. Parkin, Eur. J. Inorg. Chem., 2006, 1255 CrossRef CAS
.
- E. S. Peters, C. J. Carmalt, I. P. Parkin and D. A. Tocher, Eur. J. Inorg. Chem., 2005, 4179 CrossRef CAS
; C. J. Carmalt, E. S. Peters, I. P. Parkin, T. D. Manning and A. L. Hector, Eur. J. Inorg. Chem., 2004, 4470 CrossRef
.
- S. L. Benjamin, Y.-P. Chang, C. Gurnani, A. L. Hector, M. Huggon, W. Levason and G. Reid, Dalton Trans., 2014, 43, 16640 RSC
.
- S. L. Benjamin, Y.-P. Chang, M. Huggon, W. Levason and G. Reid, Polyhedron, 2015, 99, 230 CrossRef CAS
.
- Y.-P. Chang, W. Levason, M. E. Light and G. Reid, Dalton Trans., 2016, 45, 16262 RSC
.
- D. A. Rice, Coord. Chem. Rev., 1978, 25, 199 CrossRef CAS
; M. J. Atherton and J. H. Holloway, Adv. Inorg. Chem. Radiochem., 1979, 22, 171 CrossRef
.
-
Comprehensive Coordination Chemistry II, ed. T. J. Meyer and J. A. McCleverty, Elsevier, Oxford, 2004, vol. 4 and 5 Search PubMed
.
- U. Müller and P. Klingelhöfer, Z. Anorg. Allg. Chem., 1984, 510, 109 CrossRef
.
- M. G. B. Drew and R. J. Hobson, Inorg. Chim. Acta, 1983, 72, 233 CrossRef CAS
.
- K. Behzadi, A. G. Baghlaf and A. Thomson, J. Less-Common Met., 1987, 128, 195 CrossRef CAS
.
- V. C. Gibson, A. Shaw and D. N. Williams, Polyhedron, 1989, 8, 549 CrossRef CAS
.
- M. G. B. Drew, D. A. Rice and D. M. Williams, J. Chem. Soc., Dalton Trans., 1984, 1087 RSC
; M. G. B. Drew, D. A. Rice and D. M. Williams, Acta Crystallogr., 1980, C40, 1547 Search PubMed
.
- M. G. B. Drew, D. A. Rice and D. M. Williams, J. Chem. Soc., Dalton Trans., 1983, 2251 RSC
.
- I. Nowak, E. M. Page, D. A. Rice, A. D. Richardson, R. J. French, K. Hedberg and J. S. Ogden, Inorg. Chem., 2003, 42, 1296 CrossRef CAS PubMed
.
- G. W. A. Fowles, R. J. Hobson, D. A. Rice and K. J. Shanton, J. Chem. Soc., Chem. Commun., 1976, 552 RSC
.
-
93Nb, 100%, I = 9/2, Ξ = 24.44, Rc = 2740, Q = −0.2 × 10−28 m2.
J. Emsley, The Elements, Oxford, 1989 Search PubMed
.
- V. P. Tarasov, S. M. Sinitsyna, V. D. Kopanev, V. G. Khleboradov and Y. A. Buslaev, Koord. Khim., 1980, 6, 1568 CAS
.
- W. Levason, M. Jura, R. Ratnani, G. Reid and M. Webster, Dalton Trans., 2010, 49, 883 Search PubMed
.
- F. R. Hartley, S. G. Murray, W. Levason, H. E. Soutter and C. A. McAuliffe, Inorg. Chim. Acta, 1979, 35, 265 CrossRef CAS
.
- D. J. Gulliver, E. G. Hope, W. Levason, S. G. Murray, D. M. Potter and G. L. Marshall, J. Chem. Soc., Perkin Trans. 2, 1984, 429 RSC
.
- B. Morosin, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1974, 30, 551 CrossRef CAS
.
- B. E. Brown and D. J. Beerntsen, Acta Crystallogr., 1965, 18, 31 CrossRef CAS
.
- G. M. Sheldrick, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 2015, 71, 3 CrossRef PubMed
.
-
CrysAlis PRO, Agilent Technologies Ltd., Yarnton, Oxfordshire, England Search PubMed
.
- S. Grazulis, D. Chateigner, R. T. Downs, A. F. Yokochi, M. Quiros, L. Lutterotti, E. Manakova, J. Butkus, P. Moeck and A. Le Bail, J. Appl. Crystallogr., 2009, 42, 726 CrossRef CAS PubMed
.
- ICSD: Inorganic Crystal Structure Database (ICSD), Fachinformationszentrum Karlsruhe (FIZ), accessed via the United Kingdom Chemical Database Service, D. A. Fletcher, R. F. McMeeking and D. J. Parkin, Chem. Inf. Comput. Sci., 1996, 36, 746 CrossRef CAS
.
Footnote |
| † Electronic supplementary information (ESI) available: Crystallographic parameters (Table S1), XRD data (Fig. S1) and SEM images (Fig. S2) for the NbS2 film obtained from [NbSCl3{nBuS(CH2)3SnBu}] and XRD data from a NbSe2 film obtained by using less [NbSe2Cl3(SenBu2)] precursor (∼50 mg). Cif files for the four crystallographically characterised precursor complexes. CCDC 1542831–1542834. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c7dt01911d |
| This journal is © The Royal Society of Chemistry 2017 |