First-principles study of Ga-vacancy induced magnetism in β-Ga2O3†
Abstract
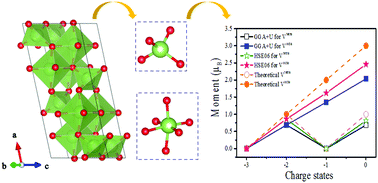
First principles calculations based on density functional theory were performed to study the electronic structure and magnetic properties of β-Ga2O3 in the presence of cation vacancies. We investigated two kinds of Ga vacancies at different symmetry sites and the consequent structural distortion and defect states. We found that both the six-fold coordinated octahedral site and the four-fold coordinated tetrahedral site vacancies can lead to a spin polarized ground state. Furthermore, the calculation identified a relationship between the spin polarization and the charge states of the vacancies, which might be explained by a molecular orbital model consisting of uncompensated O2− 2p dangling bonds. The calculations for the two vacancy systems also indicated a potential long-range ferromagnetic order which is beneficial for spintronics application.


 Please wait while we load your content...
Please wait while we load your content...