Control of pH for separated quantitation of nitrite and cyanide ions using photoluminescent copper nanoclusters†
Abstract
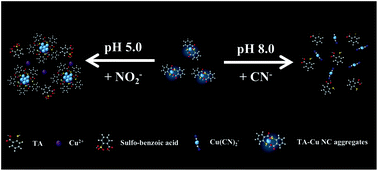
A dual sensing probe has been developed for the detection of nitrite (NO2−) and cyanide (CN−) ions based on the analyte-induced photoluminescence (PL) quenching of thiosalicylic acid (TA) capped-copper (Cu) nanocluster (NC) aggregates. The TA–Cu NC aggregates prepared from Cu2+ and TA emit at 420 nm when excited at 338 nm, with a quantum yield of 13.2%. The PL quenching of the TA–Cu NC aggregates by NO2− is through a redox reaction between HNO2 and TA under acidic conditions, while that by CN− ions in a basic solution is through an etching process. By controlling pH values at 5.0 and then at 8.0, the probe allows consecutive quantitation of NO2− and CN− ions in water samples, with limits of detection of 5 μM and 5 nM, respectively, at a signal-to-noise ratio of 3. The practicality of this probe has been validated through the determination of the concentrations of NO2− and CN− ions in representative lake water samples.


 Please wait while we load your content...
Please wait while we load your content...