A novel electron carrier molecule based on a viologen derivative for visible light-driven CO2 reduction to formic acid with the system of zinc porphyrin and formate dehydrogenase
S.
Ikeyama
a and
Y.
Amao
 *ab
*ab
aThe Advanced Research Institute for Natural Science and Technology, Osaka City University, 3-3-138 Sugimoto, Sumiyoshi-ku, Osaka 558-8585, Japan. E-mail: amao@ocarina.osaka-cu.ac.jp
bResearch Centre for Artificial Photosynthesis (ReCAP), Osaka City University, 3-3-138 Sugimoto, Sumiyoshi-ku, Osaka 558-8585, Japan
First published on 25th July 2017
Abstract
To develop a visible-light driven conversion of CO2 to formic acid system consisting of water-soluble zinc porphyrin, an electron carrier and formate dehydrogenase (FDH) in the presence of triethanolamine (TEOA) as an electron donor, 1,1′-diaminoethyl-4,4′-bipyridinium salt (DAV) as a novel electron carrier with high affinity for FDH was applied.
A photoredox system consisting of an electron donor, a visible-light sensitizer, an electron carrier and a catalyst is widely used for the conversion of visible-light energy to chemical energy.1–12 This system is a simplification of the photoinduced electron transfer process in natural photosynthesis processes. By using a reduced electron carrier as a substrate or a co-enzyme for the catalyst or biocatalyst in this system, hydrogen production and CO2 reduction systems are developed. For visible light-driven hydrogen production using the photoredox system, platinum nano-particles or biocatalyst hydrogenase has been used as a catalyst for hydrogen production with visible-light sensitizers such as water soluble zinc porphyrins, ruthenium polypyridyl coordination complexes and chlorophyll-a.13–19 The visible-light driven chemical conversion of organic compounds has also been developed using this photoredox system in the presence of various NAD+-dependent dehydrogenases such as lactate (LDH), formate (FDH), aldehyde (AldDH) and alcohol (ADH) dehydrogenase for the chemical conversion of valuable organic compounds such as lactic acid,20 ethanol,21 formic acid,22–31 methanol,32–34etc. Among these dehydrogenases, FDH catalyzes the conversion of CO2 to formic acid in the presence of a suitable electron carrier as a co-enzyme such as NADH, or one electron-reduced bipyridinium salts (BPs). Thus, a visible-light driven CO2 conversion to formic acid system is developed with the combination of the photoredox system and FDH. As FDH catalyzes the oxidation and reduction between formic acid and CO2 with the redox coupling of NAD+/NADH, for example, CO2 conversion to formic acid is developed with the photoredox system consisting of an electron donor, a visible-light sensitizer, and NAD+ as an electron carrier in the presence of FDH.35–40 There is also research being conducted to genetically modify FDH as a catalyst suitable for formic acid production from CO2.41 However, the NAD dimer, that is an inactive co-enzyme for NAD+-dependent dehydrogenases, is formed between the one-electron reduced form of NAD+s with a visible-light sensitizer such as tris(bipyridine)ruthenium(II), Ru(bpy)32+.42 Thus, it is quite difficult to achieve the photoredox system based on the combination of NAD+ photoreduction and FDH for the conversion of CO2 to formic acid. Even if the photoreduction of NADH with the photoredox system could be achieved, NAD+ is a very expensive reagent, and it is necessary to improve its usage and turnover of the redox coupling of NAD+/NADH. As the produced NADH acts as an electron donor (sacrificial reagent) and is consumed in the photoredox system, the redox coupling of NAD+/NADH is not suitable to use for this system.
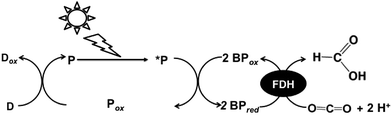
On the other hand, visible-light driven CO2 conversion to formic acid also is developed with the photoredox system consisting of an electron donor (D), a visible-light sensitizer (P), and a simple electron carrier, bipyridinium salts (BPs), in the presence of FDH as shown in Fig. 1.5,43
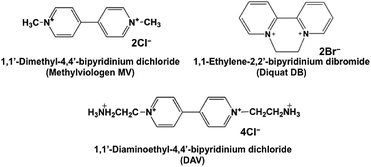
As the BPs are easily reduced by a visible-light sensitizer in this system, more effective visible-light driven CO2 conversion to formic acid with BPs will be developed compared with that of the system using an NAD+/NADH redox couple in the visible light-driven photoredox system. The visible light-driven photoredox system for formic acid production from CO2 (hydrogen carbonate ion) with FDH and typical 4,4′-bipyridinium salt (4,4′-BP), methylviologen (MV) photoreduction with a system containing Ru(bpy)32+ as a visible-light sensitizer and mercaptoethanol (RSH) as an electron donor has been reported for the first time.22–26 In contrast, we previously reported the visible light-driven photoredox system for formic acid production from CO2 or hydrogen carbonate ions with the system consisting of MV, FDH and water-soluble zinc porphyrin27–29 or chlorophyll-a30 in the presence of triethanol amine (TEOA) as an electron donor. To improve the yield of formic acid from CO2 in this system, an effective electron carrier based on the bipyridinium salt for activation of FDH for CO2 to formic acid conversion is required. For example, 1,1′-dialkyl-2,2′-bipyridinium salt derivatives (2,2′-BPs) act as the effective electron carrier for the formic acid production from CO2 in the visible light-driven redox system with FDH instead of MV. By using 2,2′-BPs, the redox potential and dihedral angle between pyridine rings will be controlled by the methylene chain length bridged between pyridine rings in bipyridinium salt. As 1,1′-ethylene-2,2′-bipyridinium (DB; chemical structure: Fig. 2) is easy to reduce compared with the other 2,2′-BPs and its reduced form has a small dihedral angle, effective formic acid production is observed by using DB in the system using water-soluble zinc porphyrin as a visible-light sensitizer.31,44,45
We also have reported an enzymatic kinetic analysis on the formic acid production from CO2 with FDH in the presence of various reduced forms of 4,4′-BP. Using 1,1′-diaminoethyl-4,4′-bipyridinium dichloride (DAV; chemical structure: Fig. 2), we discovered the effective reduction of CO2 to formic acid with FDH. The catalytic efficiency of FDH with the reduced form of DAV was estimated to be more than 560 times larger than that of the natural co-enzyme NADH.46,47 Thus, the DAV is the effective electron carrier for the visible light-driven photoredox system for formic acid production from CO2 with the system consisting of FDH and water-soluble zinc porphyrin in the presence of TEOA. In this communication, effective electron carrier DAV is applied to the visible-light driven conversion of CO2 to formic acid with the system consisting of water-soluble zinc tetraphenylporphyrin tetrasulfonate (ZnTPPS) and FDH in the presence of TEOA.
FDH from Candida boidinii (MW. 74 kDa) was purchased from Roche Diagnostics K.K. MV and tetraphenylporphyrin tetrasulfonate (H2TPPS) were purchased from Tokyo Kasei Co. Ltd. DB was purchased from WAKO Chemicals Co. Ltd. The other chemical regents were of analytical grade or the highest grade available.
ZnTPPS was synthesized by refluxing H2TPPS with about 10 times molar equivalent of zinc acetate in 100 ml of methanol at 40 °C for 2 h according to the previous report.31–34
DAV was synthesized according to the previous report.46,47
A solution consisting of TEOA (0.3 M), ZnTPPS (10 μM) and BP (100 μM) in 5.0 ml of 10 mM sodium pyrophosphate buffer (pH 7.4) was deaerated by freeze–pump–thaw cycles repeated 5 times and then was irradiated with a 250 W halogen lamp (TOSHIBA JD110V215WNP-EH-TB) at a distance of 5.0 cm at 30 °C. The concentration of the reduced form of BP was estimated by using a UV-Vis absorption spectrum (Shimadzu Multispec-1500). Molar coefficients for reduced forms of DAV, MV and DB were estimated to be 4200 (λmax: 605 nm), 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (λmax: 605 nm) and 4313.5 M−1 cm−1 (λmax: 450 nm), respectively.31,44–47 When the solution consisting of TEOA, ZnTPPS and BP was irradiated with visible light, the absorbance attributed to the absorption band of the reduced form of BP increased with irradiation time. Fig. 3 shows the time dependence of the reduced form of BP production.
000 (λmax: 605 nm) and 4313.5 M−1 cm−1 (λmax: 450 nm), respectively.31,44–47 When the solution consisting of TEOA, ZnTPPS and BP was irradiated with visible light, the absorbance attributed to the absorption band of the reduced form of BP increased with irradiation time. Fig. 3 shows the time dependence of the reduced form of BP production.
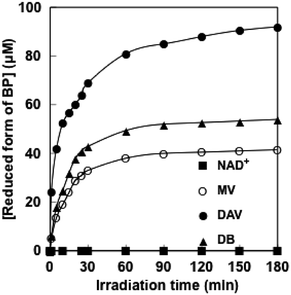 | ||
| Fig. 3 Time dependence of reduced forms of DAV (●), MV (○), DB (▲) and NAD+ (■) production under steady state irradiation with visible light using a 250 W halogen lamp. | ||
From the result in Fig. 3, it can be seen that the reduced form of BP was produced with increasing the irradiation time. The initial rate was estimated from the gradient of the linear part of the reduced form of BP production within 10 min irradiation. The initial rates for the reduced form of DAV, MV and DB production were estimated to be 5.2, 1.9 and 2.5 μM min−1, respectively. After 180 min irradiation, the concentration of reduced forms of DAV, MV and DB was 92.0, 41.5 and 54.0 μM, respectively. The error values of the photoreduction of DAV, MV and DB in the five experiments after 10 and 180 min irradiation were estimated within 0.1 and 1.0 μM, respectively. After 180 min irradiation, the conversion yields of reduced forms of DAV, MV and DB production from the oxidized form of each BP were estimated to be 0.920, 0.415 and 0.540, respectively. The DAV was almost completely reduced with the photosensitization of ZnTPPS after 180 min light irradiation. When the solution consisting of TEOA, ZnTPPS and NAD+ was irradiated with visible light, in contrast, no NADH was produced with increasing the irradiation time. Moreover, the one-electron reduction of NAD+ also did not proceed. In order to reduce NAD+ to NADH, two electrons were required. In the case of NAD+ reduction, direct two-electron reduction of NAD+ with the sensitization of ZnTPPS did not occur. The first step is the one-electron reduction of NAD+ and then two-electron reduction of NAD+ occurred as a second step. However, even if a one-electron reduced form of NAD+ was formed, dimerization (NAD dimer) occurred between the one-electron reduced form of NAD+s. As the NAD dimer was an inactive co-enzyme for FDH, thus, NAD+ photoreduction is not suitable for the photoredox system with FDH.
For the intermolecular interaction between ZnTPPS and MV or DB, two positively charged pyridinium moieties in MV or DB were interacted with two neighbouring sulfo-groups in ZnTPPS by the electrostatic effect. Thus, MV or DB was located outside the porphyrin ring. For the intermolecular interaction between ZnTPPS and DAV, on the other hand, two amino groups and two positively charged pyridinium moieties in DAV were strongly interacted with a sulfo-group in ZnTPPS by the electrostatic effect. Thus, the DAV strongly interacts with ZnTPPS at lower concentrations than that of MV or DB. Thus, the efficiency of photoreduction of DAV with ZnTPPS was increased compared with that of MV or DB.
A solution consisting of TEOA (0.3 M), ZnTPPS (10 μM), BP (100 μM) and FDH (6.4 μM) in 5.0 ml of 10 mM sodium pyrophosphate buffer (pH 7.4) was deaerated by freeze–pump–thaw cycles repeated 5 times and the gas phase was replaced with CO2. The sample solution was irradiated with a 250 W halogen lamp at 30 °C. The amount of formic acid was detected by ion chromatography (Dionex ICS-1100). When the reaction mixture consisting of TEOA, ZnTPPS, BP and FDH in the CO2 saturated sodium pyrophosphate buffer was irradiated with visible light at 30 °C, formic acid production was observed with irradiation time. As shown in Fig. 4, the formic acid production after 1 h irradiation with DAV, MV and DB in the CO2 saturated solution consisting of TEOA, ZnTPPS and FDH was 120, 60.5 and 70.0 μMh−1, respectively. When the solution consisting of TEOA, ZnTPPS, NAD+ and FDH in the CO2 saturated buffer was irradiated with visible light, in contrast, no formic acid was produced with increasing the irradiation time. As no NADH was produced with the photosensitization of ZnTPPS, CO2 reduction with FDH did not proceed.
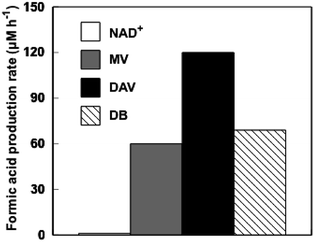 | ||
| Fig. 4 The formic acid production after 1 h irradiation with DAV, MV and DB in the CO2 saturated solution consisting of TEOA, ZnTPPS and FDH. | ||
For the system consisting of TEOA, ZnTPPS, DAV and FDH, the amount of formic acid production was estimated to be 0.6 μmol after 1 h irradiation. This indicated that 1.2 μmol of proton and 0.6 μmol of CO2, that were 24 times the amount of ZnTPPS (50 nmol) in the sample solution, were reduced to formic acid molecules. For the system consisting of TEOA, ZnTPPS, MV and FDH, on the other hand, the amount of formic acid production was 0.3 μmol after 1 h irradiation, the turnover number of ZnTPPS was estimated to be 12 h−1. By using DB as an electron carrier, the turnover number of ZnTPPS was estimated to be 14 h−1. Therefore, the ZnTPPS appeared to serve as the system for transferring electrons from TEOA to a more reductive molecule in all cases. Moreover, the turnover number of DAV, MV and DB was estimated to be 1.2, 0.6 and 0.7 h−1, respectively. The turnover number of FDH in DAV, MV and DB was estimated to be 18.8, 9.45 and 10.9 h−1, respectively. The turnover number of ZnTPPS, BP and FDH, and total formic acid production with visible light-driven CO2 reduction to formic acid with FDH and ZnTPPS are summarized in Table 1.
| BPs | TON of ZnTPPS (h−1) | TON of BP (h−1) | TON of FDH (h−1) | Total formic acid production (μM) |
|---|---|---|---|---|
| DAV | 24 | 1.2 | 18.8 | 120 |
| MV | 12 | 0.6 | 9.45 | 60.5 |
| DB | 14 | 0.7 | 10.9 | 70.0 |
Next let us focus on the reason for the improvement of CO2 photoreduction to formic acid with the system of ZnTPPS, DAV and FDH. We previously reported that the reduced form of DAV had high affinity for FDH compared with the other 4,4′-BPs such as MV. By enzyme kinetic analysis, the catalytic efficiency of the reduced form of DAV was about approximately 30 times larger than that of MV and estimated to be more than 560 times that of NADH, indicating that 4,4′-BP with an amino group has higher affinity for FDH compared with those of other 4,4′-BPs.43,46,47 However, in the visible light-driven CO2 reduction to formic acid with FDH and ZnTPPS, a formic acid production rate with DAV was only twice more than that of MV. The time dependence of formic acid production using DAV in visible light-driven CO2 reduction with ZnTPPS, FDH and TEOA was checked. The formic acid production was saturated at 1 h irradiation. By enzymatic kinetic analysis, Michaelis constants (Km) of the reduced form of DAV, MV and DB for FDH were estimated to be 17.0, 212 and 78.0 μM, respectively.46,47 By using DAV, the maximum velocity (Vmax) for formic acid production with FDH was reached at a low concentration (∼34 μM). When the concentration of DAV in the system containing ZnTPPS, FDH and TEOA was reduced to 20 μM, formic acid production linearly increased with light irradiation over 3 h. However, the formic acid production rate decreased to 21 μM h−1. As DAV was easily reduced with ZnTPPS and the reduced form of DAV was accumulated, it is considered that the reduced form of DAV acted as an inhibitor for FDH as one possibility. Details of the effect of BP on the visible light-driven CO2 reduction to formic acid with FDH and ZnTPPS are currently being investigated.
Acknowledgements
This work was partially supported by Grants-in-Aid for Challenging Exploratory Research (Japan Society for the Promotion of Science) (15K14239), and a Grant-in-Aid for Scientific Research on Innovative Areas “Artificial Photosynthesis (2406)”.Notes and references
- J. Kiwi, K. Kalyanasundaram and M. Grätzel, Struct. Bonding, 1982, 49, 37 CrossRef.
- L. A. Kelly and M. A. J. Rodgers, J. Phys. Chem., 1994, 98, 6386 CrossRef CAS.
- J. R. Darwent, P. Douglas, A. Harriman, G. Porter and M. C. Richoux, Coord. Chem. Rev., 1982, 44, 93 CrossRef.
- P. A. Brugger, P. Cuendet and M. Grätzel, J. Am. Chem. Soc., 1981, 103, 2923 CrossRef CAS.
- Y. Amao, ChemCatChem, 2011, 3, 458 CrossRef CAS.
- Y. Amao, Curr. Top. Biotechnol., 2009, 5, 71 CAS.
- I. Okura, Coord. Chem. Rev., 1985, 68, 53 CrossRef CAS.
- I. Okura, Biochimie, 1986, 68, 189 CrossRef CAS PubMed.
- J. Kiwi and M. Grätzel, J. Am. Chem. Soc., 1979, 101, 7214 CrossRef CAS.
- Y. Amao, Curr. Nanosci., 2008, 4, 45 CrossRef CAS.
- Y. Amao and I. Okura, Sensitization by Metal Complexes Towards Future Artificial Photosynthesis in Photocatalysis, Kodansha Springer, 2002 Search PubMed.
- A. Harriman, G. Porter and M.-C. Richoux, J. Chem. Soc., Faraday Trans. 2, 1981, 77, 833 RSC.
- Y. Amao and I. Okura, J. Mol. Catal. A: Chem., 1996, 105, 125 CrossRef CAS.
- Y. Amao, Y. Tomonou, Y. Ishikawa and I. Okura, Int. J. Hydrogen Energy, 2002, 27, 621 CrossRef CAS.
- T. Kamachi, T. Hiraishi and I. Okura, Chem. Lett., 1995, 24, 33 CrossRef.
- Y. Tomonou and Y. Amao, Int. J. Hydrogen Energy, 2004, 29, 159 CrossRef CAS.
- Y. Tomonou and Y. Amao, BioMetals, 2002, 15, 391 CrossRef CAS PubMed.
- Y. Tomonou and Y. Amao, BioMetals, 2003, 16, 419 CrossRef CAS PubMed.
- Y. Amao, Y. Maki and Y. Fuchino, J. Phys. Chem. C, 2009, 113, 16811 CAS.
- R. Miyatani and Y. Amao, Photochem. Photobiol. Sci., 2004, 3, 681 CAS.
- Y. Amao, N. Shuto and H. Iwakuni, Appl. Catal., B, 2016, 180, 403 CrossRef CAS.
- D. Mandler and I. Willner, J. Chem. Soc., Perkin Trans. 1, 1988, 2, 997 RSC.
- I. Willner and D. Mandler, J. Am. Chem. Soc., 1989, 111, 1330 CrossRef CAS.
- I. Willner, N. Lapidot, A. Riklin, R. Kasher, E. Zahavy and E. Katz, J. Am. Chem. Soc., 1994, 116, 1428 CrossRef CAS.
- I. Willner, I. Willner and N. Lapidot, J. Am. Chem. Soc., 1990, 112, 6438 CrossRef CAS.
- M. Kodaka and Y. Kubota, J. Chem. Soc., Perkin Trans. 1, 1999, 2, 891 RSC.
- R. Miyatani and Y. Amao, Biotechnol. Lett., 2002, 24, 1931 CrossRef CAS.
- R. Miyatani and Y. Amao, J. Mol. Catal. B: Enzym., 2004, 27, 121 CrossRef CAS.
- R. Miyatani and Y. Amao, J. Jpn. Pet. Inst., 2004, 47, 27 CrossRef CAS.
- I. Tsujisho, M. Toyoda and Y. Amao, Catal. Commun., 2006, 7, 173 CrossRef CAS.
- Y. Amao, R. Abe and S. Shiotani, J. Photochem. Photobiol., A, 2015, 313, 149 CrossRef CAS.
- Y. Amao and T. Watanabe, Chem. Lett., 2004, 33, 1544 CrossRef CAS.
- Y. Amao and T. Watanabe, Appl. Catal., B, 2009, 86, 109 CrossRef CAS.
- Y. Amao, Prepr. Pap. - Am. Chem. Soc., Div. Fuel Chem., 2010, 55, 91 CAS.
- R. K. Yadav, G. H.Oh, N.-J. Park, A. Kumar, K.-J. Kong and J.-O. Baeg, J. Am. Chem. Soc., 2014, 136, 16728 CrossRef CAS PubMed.
- R. K. Yadav, J.-O. Baeg, G. H. Oh, N.-J. Park, K.-J. Kong, J. Kim, D. W. Hwang and S. K. Biswas, J. Am. Chem. Soc., 2012, 134, 11455 CrossRef CAS PubMed.
- W. S. Choi, S.-H. Lee, J.-W. Ko and C.-B. Park, ChemSusChem, 2016, 9, 1559 CrossRef CAS PubMed.
- S. K. Kuk, R. K. Singh, D. H. Nam, R. Singh, J.-K. Lee and C.-B. Park, Angew. Chem., 2017, 129, 3885 CrossRef.
- R. Cazelles, J. Drone, F. Fajula, O. Ersen, S. Moldovan and A. Galarneau, New J. Chem., 2013, 37, 3721 RSC.
- X. Ji, Z. Su, P. Wang, G. Ma and S. Zhang, ACS Nano, 2015, 9, 4600 CrossRef CAS PubMed.
- H. Choe, J. C. Joo, D. H. Cho, M. H. Kim, S. H. Lee, K. D. Jung and Y. H. Kim, PLoS One, 2014, 9, e103111 Search PubMed.
- K. Hironaka, S. Fukuzumi and T. Tanaka, J. Chem. Soc., Perkin Trans. 2, 1984, 1705 RSC.
- Y. Amao, Chem. Lett., 2017, 46, 780 CrossRef.
- Y. Amao and S. Ikeyama, Chem. Lett., 2015, 44, 1182 CrossRef CAS.
- S. Ikeyama, R. Abe, S. Shiotani and Y. Amao, Chem. Lett., 2016, 45, 979 CrossRef.
- S. Ikeyama and Y. Amao, Chem. Lett., 2016, 45, 1259 CrossRef CAS.
- S. Ikeyama and Y. Amao, ChemCatChem, 2017, 9, 833 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2017 |