Heterocyclic bismuth(iii) compounds with transannular N→Bi interactions as catalysts for the oxidation of thiophenol to diphenyldisulfide†
Abstract
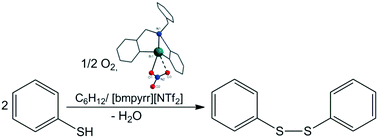
The reactions between the diorganobismuth(III) bromides [RCH2N(CH2C6H4)2]BiBr [R = C6H5 (1), C6H5CH2 (2)] and appropriate silver salts resulted in new diorganobismuth(III) compounds of the general formula [RCH2N(CH2C6H4)2]BiX [R = C6H5, X = ONO2 (3), OSO2CF3 (4), OSO2C6H4 (CHCH2)-4 (5); R = C6H5CH2, X = ONO2 (6)], based on a butterfly-like tetrahydro-dibenzo[c,f][1,5]azabismocine heterocyclic framework. The new species were structurally characterized in solution by 1H, 13C{H} and 19F{H} NMR and in the solid state by IR spectroscopy and single-crystal X-ray diffraction. The nitrogen atom is intramolecularly coordinated to bismuth, thus resulting in hypercoordinated species of type 12-Bi-5 (3 and 6) and 10-Bi-4 (4 and 5). In addition, compound 4 shows bismuth⋯π arene and compounds 3 and 6 bismuth⋯oxygen intermolecular interactions, thus leading to dimers in the solid state. These compounds were investigated as catalysts for the oxidation of thiophenol to diphenyl disulfide by using air as an oxidizing agent, both in cyclohexane and in an ionic liquid (1-butyl-1-methylpyrrolidinium bis(trifluoromethanesulfonyl)imide), at temperatures below 100 °C, affording high reaction rates (TON 34.8, with 100% conversion after 5 h) and a total selectivity to the targeted product.
- This article is part of the themed collection: Catalytic reactivity of surfaces: in recognition of François Gault


 Please wait while we load your content...
Please wait while we load your content...