One-step facile fabrication of controllable microcone and micromolar silicon arrays with tunable wettability by liquid-assisted femtosecond laser irradiation†
Abstract
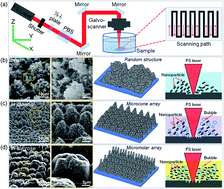
Micro/nanostructured silicon surfaces are attracting more and more research attention because of the wide range of applications in optoelectronic devices, microelectronics, microfluidics, and biomedical devices. Despite numerous efforts for fabricating a variety of micro/nanostructures, a one-step, facile and effective method for preparing diverse, three-dimensional micro/nanostructures is still desired. In this paper, a new approach based on liquid (ethanol and sucrose solution) assisted femtosecond laser irradiation on silicon substrates was proposed for the preparation of controllable microcones and micromolars arrays. Their height can be controlled from 3.3 to 17.6 μm for microcones and 5.9∼33.7 μm for micromolars by adjusting the pulse energy. The processed surfaces are superhydrophilic (25.05∼2.46°), superoleophilic (7.22∼0°), and underwater superoleophobic (124.9∼169.2°). The surfaces further demonstrate many distinct functions such as fog collecting and volatilizing, droplet storage and transportation, and liquid directional transfer. Our proposed method features rapidness, simplicity and easiness of large-area fabrication, which may find enormous potential applications in many fields such as microfluidic devices, fluid microreactors, biomedicine, and chemical–biological sensors.

 Please wait while we load your content...
Please wait while we load your content...