Effects of the i-motif DNA loop on the fluorescence of silver nanoclusters†
Abstract
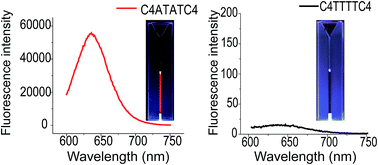
Cytosine-rich DNA strands, due to their distinct i-motif structure, are often used as stabilization templates for the formation of silver nanoclusters (Ag NCs). DNA i-motif conformation and stability are largely determined by the DNA loop region. Therefore, the optimal loop sequence contributes to the DNA template, forming a stable i-motif structure, thus promoting the formation of Ag NCs. In the present work, we prepared Ag NCs stabilized by a set of DNA sequences CCCC-X1X2X3X4-CCCC (C denotes cytosine, X denotes adenine or thymine) and investigated the loop sequence dependence of the fluorescence quantum yields (QYs) of Ag NCs. In all of the template DNA, the sequence 5′-CCCC-ATAT-CCCC-3′ was screened to be the most optimal template which stabilized clusters and could emit the brightest red fluorescence with a quantum yield (QY) of 0.78. However, for the reversed template sequence 5′-CCCC-TATA-CCCC-3′, the QY of the Ag NCs is lowered to 0.15. Such a significant difference in QYs indicates that even for the same base sequence, a different base alignment direction (starting from the 5′ or 3′ end) can have an important effect on the fluorescence of nanoclusters. Additionally, to optimize the preparation conditions of Ag NCs, the effects of reductants and pH environment on the clusters stabilized by the template sequence 5′-CCCC-ATAT-CCCC-3′ were further examined by using CD, UV and fluorescence spectroscopy, respectively.
- This article is part of the themed collection: Chemistry for Medicine: Special Collection for RSC Advances

 Please wait while we load your content...
Please wait while we load your content...